Investigation / Researchs
Files > Volume 1 > Vol 1 No 2 2016 > Investigaciones
Comparison of two methods with potential application in the detection of viremia produced by clinical dengue virus isolates
Comparación de dos métodos con aplicación potencial en la detección de viremia a partir de aislamientos clínicos de virus dengue
Available from: http://dx.doi.org/10.21931/RB/2016.01.02.5Jorge Martín*, Laura Lazo, Iris Valdés, Lázaro Gil, Yaremis Romero, Jorge Castro, Gerardo Guillén, Lisset Hermida
Abstract
Most clinical isolates do not form clear plaques on cell monolayers. Therefore, they can not be detected by plaque assay. Reliable alternative methods for the measurement of viremia are thus required for the validation of the protective efficacy of dengue vaccine candidates against circulating virus strains. In this work, two different cell culture formats (25 cm2 flasks and 24-well plates) were used to isolate dengue virus from 48 serum samples collected from monkeys. Only 8 dengue positive sera were detected after isolation in 24-well plates. In contrast, the 25 cm2-flask format allowed the detection of virus in 22 serum samples, suggesting that the Vero cell monolayer area influences virus isolation. We also compared the sensitivity of ELISA and flow cytometry for detecting dengue virus after isolation in Vero cell culture. Both techniques showed 100% specificity and similar sensitivity. In addition, there was a high agreement rate and correlation between the two techniques. Our findings suggest that ELISA and flow cytometry have similar capacity to detect and quantify dengue virus from monkey serum samples after virus isolation in cell culture.
Keywords: dengue, viremia, ELISA, flow cytometry.
Resumen
La mayoría de los asilamientos clínicos no forman placas distinguibles en las monocapas celulares, por lo que no pueden ser detectados con el método de formación de placas. Por tanto, se requieren métodos de medición de viremia alternativos y confiables para la evaluación de la capacidad protectora de candidatos vacunales contra el dengue frente a las cepas virales circulantes. En este trabajo se utilizaron dos formatos para el aislamiento de virus dengue a partir de 48 muestras de suero de mono: placas de 24 pozos y frascos de 25 cm2. Solo 8 sueros se detectaron como positivos tras el aislamiento en placas de 24 pozos, mientras que en frascos de 25 cm2 se detectaron 22 sueros positivos, lo que sugiere que el área de la monocapa celular influye en los resultados de la detección. Además, se comparó la sensibilidad del ELISA y la citometría de flujo para la detección de virus dengue tras el aislamiento en células Vero. Ambas técnicas mostraron 100% de especificidad y similar sensibilidad, además de una elevada concordancia y correlación entre los resultados de detección obtenidos con ellas. Los resultados de este trabajo sugieren que el ELISA y la citometría de flujo tienen similar capacidad para la detección y cuantificación de virus dengue a partir de sueros de monos tras el aislamiento en cultivo celular.
Palabras clave: dengue, viremia, ELISA, citometría de flujo.
Introduction
Dengue is a single-stranded positive-sense RNA virus that belongs to the Flavivirus genus in the Flaviviridae family. There are four serotypes of antigenically related dengue viruses, and infection with any of them can produce a spectrum of diseases that range from asymptomatic or mild febrile illness to the more severe dengue hemorrhagic fever and/or dengue shock syndrome 1.
Dengue is currently a major global health problem. It is estimated that 390 million DV infections occur annually in the world, of which 96 million are apparent 2. This pathogen is endemic in over 100 countries from the tropical and subtropical regions where 2.5-3.6 billion people are at risk for infection. Although many efforts have been made, there is no effective vaccine to prevent this disease. The absence of an animal model that reproduces the signs observed in humans is one of the major pitfalls. So far, primates are considered the most appropriate animal model and the effectiveness of a vaccine candidate is only assessed by the lack of viremia in vaccinated monkeys 3. Therefore, reliable measurements of viremia are of great importance in the process of validating any vaccine.
The plaque assay, based on the formation of viral plaques on cell culture, is the "gold-standard" method for the detection and titration of dengue virus 4. However, some dengue virus strains, especially among primary clinical isolates, do not form clear plaques on cell monolayers 5-7. On the other hand, the use of these isolates in challenge experiments with non-human primates may be of particular importance to clearly determine the protective capacity of vaccine candidates against circulating dengue virus strains in endemic areas.
In this work we compared two different cell culture formats to isolate virus from non-human primate serum samples using Vero cells. We also performed a preliminary comparison of two dengue virus detection methods as potential alternatives to the plaque assay for the detection and titration of viral strains that do not produce plaques on cell monolayers. Accordingly, we evaluated the sensitivity and specificity of enzyme-linked immunosorbent assay (ELISA) and flow cytometry (FC) for virus detection from infected Vero cells, and determined the correlation of viremia quantification between these two techniques.
Methods
Cell lines, virus strains and s erum samples
African green monkey kidney (Vero) cells were obtained from the National Institute for Biological Standards and Control (NIBSC accession number: 011038) and maintained in RPMI 1640 medium (Sigma Aldrich, St Louis, MO, USA) supplemented with 100 U ml−1 penicillin, 100 μgml −1 streptomycin (Gibco, Paisley, UK), 2mM glutamine (Gibco) and 5% fetal bovine serum. Baby hamster kidney (BHK-21) cells, clone 15, were obtained from the cell culture department of the Tropical Medicine Institute, Cuba, and grown under standard conditions (minimal essential medium supplemented with 5% FBS, 100 U ml−1 penicillin, 100 μgml−1 streptomycin (Gibco, Paisley, UK)). Dengue-2 virus (strain SB8553) was used as positive control in ELISA, plaque formation and flow cytometry. Serum samples were obtained from 16 infected green monkeys ( Chlorocebus aethiops sabaeus) during the first 10 days after infection with dengue-2 virus (SB8553), including the day of challenge.
Virus isolation
Sera were inoculated onto Vero cell monolayers either in 25 cm2 flasks (Costar, Cambridge, MA, USA) or in 24-well plates (Costar, Cambridge, MA, USA). A volume of 0.15 mL of serum was added to each well of flask. After one hour of incubation at 37°C, 3 mL and 1 mL of RPMI medium was added without rinsing to the 25 cm2 flasks and the 24-well plates respectively, and cultures were incubated for 6 days. Then infected cells and culture supernatants were harvested separately and assayed for the presence of virus.
ELISA
Ninety-six-well flat-bottom plates (Costar, Cambridge, MA, USA) were coated with 0.1 mL/well of a mixture of anti-dengue human (IgG) (5 μg/mL) in coating buffer (0.16% Na2CO3, 0.29% NaHCO3, pH 9.5) for 2 h at 37°C. Then plates were blocked with 0.15 mL/well of 5% skimmed milk and incubated for another 1h at 37°C. After two washes with phosphate-buffered saline-Tween 0.05% (PBS-T), 0.1 mL of culture supernatant was added in triplicate to each well and plates were incubated for 2 h at 4°C. Three additional washes were performed and then 0.1 mL/well of polyclonal hyperimmune mouse ascitic fluid, diluted 1:2 000 in PBS-T was added to the plates. After 1h of incubation at 37°C, plates were washed again three times and then 0.1 mL/well of goat anti-mouse IgG horseradish peroxidase conjugate (Sigma Aldrich, St Louis, MO, USA) was added at a dilution of 1:35 000 in PBS-T. Plates were incubated 1 h at 37°C, washed three times, and substrate solution was added. The reaction was stopped 30 min later by addition of 12.5% H2SO 4 and the optical density was measured at 492 nm (OD492nm) using an automated ELISA reader. A value of OD of two-fold the OD of the supernatant from an uninfected culture was considered as positive.
Plaque formation in BHK-21 cell cultures
Virus replication was determined by plaque formation in BHK-21 cells, according to the method of Morens et al 8. Briefly, Costar 24-well plates were inoculated with culture supernatant and incubated 4 h at 37°C in a 5% CO2 atmosphere. Later, 0.5 mL of 3% medium-viscosity carboxymethylcellulose was added. Plates were incubated at 37°C during 5 days in a 5% CO2 atmosphere. To visualize the viral plaques the monolayer was dyed with naphtol blue black solution (0.1% naphtol blue black, 0.2 M sodium acetate, 6% acetic acid).
Detection of infected cells by flow cytometry
Vero cells were removed from the flask using a cell scraper and distributed into triplicate tubes at 106 cells per tube. The cells were fixed using 200 µl of 3% formaldehyde in phosphate-buffered saline (pH 7.2) (PBS) for 15 min and 200 µl of 0.1% Triton X-100 was employed to permeabilize fixed cells. After washing with PBS, the cells were incubated with the monoclonal antibody 4G2 for 30 min at 4°C. Then cells were washed and incubated with fluorescein isothiocyanate (FITC)-labeled anti-mouse IgG for 30 min at 4°C. After incubation cells were washed again, suspended in PBS and then analyzed using a FACScan flow cytometer PASIII (Partec GmbH, Münster, Germany). Non-infected cells were used as negative control and cells infected with dengue-2 virus (strain SB8553) at 0.001 multiplicity of infection, were used as positive control. A value of three fold the value of non-infected cells was considered as positive.
Statistical analysis
The comparison between results on detection of virus by different techniques is reported in terms of agreement rates, sensitivity and specificity. The agreement rate is defined as the ratio between the number of samples equally detected (either positive or negative) by both techniques and the total number of samples analyzed. The sensitivity is the ratio of sera detected as positive by a technique with respect to the gold standard technique. The specificity is a measure of the rate of negatives that are correctly identified.
Two approaches were used for a quantitative comparison of viremia levels measured by ELISA and flow cytometry: a) the correlation between OD 492nm and percentage of infected cells, assessed by the Pearson´s correlation coefficient and b) the linear regression test. Both analyses were carried out with the GraphPad Prism version 5.00 for Windows (GraphPad Software, San Diego, CA, USA).
Results and Discussion
Comparison of two cell culture formats for virus isolation in Vero cells
Four methods of virus isolation have been routinely used for dengue viruses: intracerebral inoculation of newborn mice, intrathoracic inoculation of adult mosquitoes, inoculation of mosquito cell cultures and inoculation of mammalian cell cultures 9. Specifically for sera collected from monkeys, the most commonly used system is the mammalian cell culture. However, it is unclear how the number of cells in the monolayer influences the results. Generally, 25 cm2 flasks have been the preferred format 10-12 although 24-well plates could offer the advantages of handling a higher number of samples and cost reduction.
We performed a virus isolation experiment using both cell culture formats and 48 serum samples from green monkeys. Samples were collected during the first 10 days after dengue virus infection, including the day of challenge, with the aim of having samples with expected low and high viral loads. The virus isolated was detected by plaque formation (PF) in BHK-21 cells (Table 1).
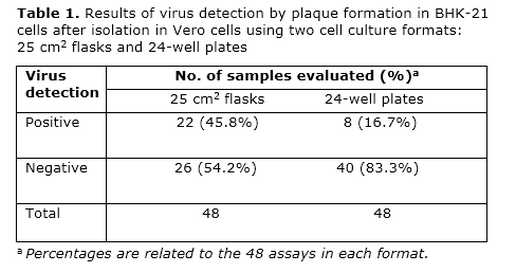
Surprisingly, only 8 (16.7%) sera were detected as positive after isolation using 24-well plates. In contrast, the use of 25 cm2 flasks led to the detection of 22 (45.8%) positive sera after isolation, including the samples detected with the 24-well format. This result points to the importance of the cell monolayer area in the isolation system for the subsequent viral detection. Therefore, a thorough study on the optimal area of the cell monolayer should be carried out in the future, looking for a balance between higher sensitivity, maneuverability and cost.
Comparison of ELISA and flow cytometry for dengue virus detection and quantification
We also compared the sensitivity and specificity of ELISA and flow cytometry for the detection of dengue virus in a set of 187 serum samples collected from monkeys. Based on the results of the previous section, virus isolation was performed using Vero cell culture in 25 cm2 flasks. The cells from each flask were harvested for FC analysis while the culture supernatant was collected for evaluation by ELISA and plaque formation in BHK-21 cells. The plaque formation assay was used as a control.
Table 2 shows the number of sera detected as positive or negative by ELISA and FC. Both techniques showed 100% specificity, with no false positive detections, and similar sensitivity (84.2% for ELISA versus 78.9% for FC). In general, the agreement rate between both techniques was 97.3% (182/187) which indicates that the two methods are approximately equal regarding their capacity to detect dengue virus.

The validation of dengue vaccine candidates in a non-human primate model relies not only on the detection of virus in serum, but also on its quantification. Therefore, having a quantitative method to analyze viremia in monkeys, in addition to highly sensitive isolation systems, is of paramount importance for vaccinologists. The two methods used in this work provide a measure of the amount of virus present in serum samples. In the case of flow cytometry, infective viral particles are quantified through the percentage of infected cells. In turn, ELISA allows determining the level of virus present in the culture supernatant, independently of its viability.
Figure 1A shows a linear regression between the levels of viremia detected by both techniques for a data set of 112 serum samples. The significant linear relation (r2=0.9181, p<0.0001) indicates that, in addition to the similar dengue virus detection rates, ELISA and flow cytometry yielded similar quantification patterns. Figure 1B illustrates this fact with three representative animals. These monkeys were selected from those with at least one day of positive detection and are representative of the minimum (0.6821), average (0.9441) and maximum (0.9942) correlation coefficients.
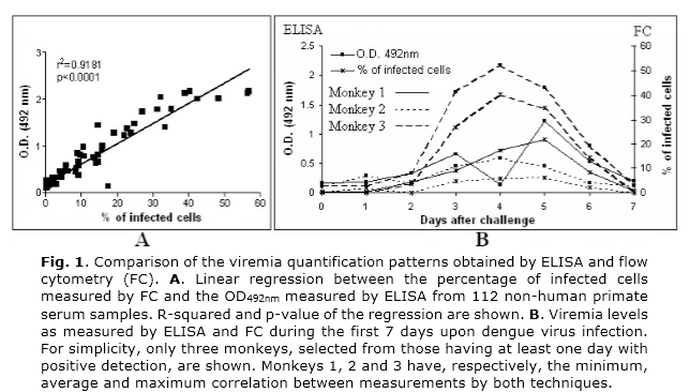
To our knowledge, ELISA has not been used for detecting and quantifying dengue virus. Since ELISA is a cheap and simple technique, these results justify its potential application in dengue viremia studies beyond its current employment in serological assessment. In turn, FC has been used to detect dengue virus in different cell lines 1 and a flow cytometry-based assay for measuring dengue virus neutralization has been reported recently 13. However, FC has not been used before to quantify viremia in monkey serum samples. Therefore, our results provide evidences for the first time that these techniques can be reliably used for the detection and quantification of viremia in non-human primates.
Although plaque assay is the gold-standard for the detection and quantification of viremia in non-human primate models and is a highly sensitive method, most clinical isolates do not produce distinct plaques on cell culture monolayers. This poses a problem for the evaluation of the protective efficacy of vaccine candidates against circulating dengue virus strains. RT-PCR is another highly sensitive method that can be used for this purpose 4, but it may amplify plasmatic viral genomes from non-infective immune complexes or viral fragments, leading to misinterpretation of results. Since ELISA is a simple, economic and time-saving method, our findings support its potential use for dengue virus detection and quantification after viral isolation in cell culture.
In summary, here we report a preliminary study on the performance of methods for the detection of viremia in monkeys infected with dengue virus. We found evidence that the area of the cell monolayer in different cell culture formats may influence the results of subsequent virus detection. Moreover, we demonstrated that flow cytometry and ELISA have similar sensitivity when applied to the same serum sample. Finally, this work evidences the need for a more thorough statistically founded study of the advantages and disadvantages of the methods available for virus detection. A reliable comparison of the techniques used for the detection of viremia in monkeys is required for the scientific community to adopt a standard procedure in dengue vaccine assessment.
References
- World Health Organization. Dengue Guidelines for Diagnosis, Treatment, Prevention, and Control. 2009. Accessed 10 March 2015. Available in: http://www.who.int/rpc/guidelines/9789241547871/ en/.
- Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, et al. The global distribution and burden of dengue. Nature 2013; 496:504-7.
- Sariol CA and White LJ. Utility, limitations, and future of non-human primates for dengue research and vaccine development. Front Immunol 2014; 5:452.
- Mungrue K. The laboratory diagnosis of dengue virus infection, a review. Advance Laboratory Medicine International 2014; 4:1-8.
- Barbana V, Munoz-Jordan JL, Santiago GA, Mantela N, Girerda Y, Guliaa S, Claudea JB, Lang J. Broad neutralization of wild-type dengue virus isolates following immunization in monkeys with a tetravalent dengue vaccine based on chimeric Yellow Fever 17D/Dengue viruses. Virology 2012; 429:91-98.
- Kraus AA, Messer W, Haymore LB, de Silva AM. Comparison of Plaque- and Flow Cytometry-Based Methods for Measuring Dengue Virus Neutralization. J Clin Microbiol 2007; 45:3777-3780.
- Liu L, Wen K, Li J, Hu D, Huang Y, Qiu L, Cai J, and Che X. Comparison of Plaque- and Enzyme-Linked Immunospot-Based Assays To Measure the Neutralizing Activities of Monoclonal Antibodies Specific to Domain III of Dengue Virus Envelope Protein. Clin Vaccine Immunol 2012; 19:73-78.
- Morens DM, Halstead SB, Repik PM. Simplified plaque reduction assay for Dengue viruses by semimicro methods in BHK 21 cells: comparison of the BHK suspension test with standard plaque reduction neutralization. J Clin Microbiol 1985; 22:250-4.
- Gubler DJ. Dengue and Dengue Hemorrhagic Fever. Clin Microbiol Rev 1998; 11:480-96.
- Putnak R, Barvir DA, Burrous JM, Dubois DR, D'Andrea VM, Hoke CH et al. Development of a purified, inactivated, dengue-2 virus vaccine prototype in Vero cells: immunogenicity and protection in mice and rhesus monkeys. J Infect Dis 1996; 174:1176-84.
- Kochel TJ, Raviprakash K, Hayes CG, Watts DM, Russell KL, Gozalo AS, et al. A dengue virus serotype-1 DNA vaccine induces virus-neutralizing antibodies and provides protection from viral challenge in Aotus monkeys. Vaccine 2000; 18:3166-73.
- Raviprakash K, Ewing D, Simmons M, Porter KR, Jones TR, Hayes CG et al. Needle-free Biojector injection of a dengue virus type1 DNA vaccine with human immunostimulatory sequences and the GM-CSF gene increases immunogenicity and protection from virus challenge in Aotus monkeys. Virology 2003; 315:345-52.
- Kraus AA, Messer W, Haymore LB, de Silva AM. Comparison of plaque and flow cytometry-based methods for measuring dengue virus neutralization. J Clin Microbiol 2007; 45: 377.
Recibido: 17 de enero de 2016.
Aprobado: 23 de marzo de 2016.
Center for Genetic Engineering and Biotechnology (CIGB). Havana, Cuba.
*Corresponding author: Jorge Martín Machado. Center for Genetic Engineering and Biotechnology (CIGB). Ave 31, P.O. Box 6162, Havana 6, 10 600, Cuba.
Fax (53-7) 271 4764, Phone: (53-7) 271 6022. E-mail: [email protected]


