2016.01.04.6
Files > Volume 1 > Vol 1 No 4 2016 > Investigaciones
INVESTIGACIÓN
Quantification of polysaccharide Vi in the typhoid vaccine VaxTyViTM by sandwich ELISA using a monoclonal antibody.
Cuantificación del polisacárido Vi en la vacuna contra la fiebre tifoidea VaxTyViTM mediante ELISA en sandwich utilizando un anticuerpo monoclonal.
Available from: http://dx.doi.org/10.21931/RB/2016.01.04.6
Elizabeth González Aznar, Fátima Reyes, Nevis Amín Blanco, Oscar Otero Alfaro, Maribel Cuello Pérez, Fidel Ramírez Bencomo and Reinaldo Acevedo Groguez
_______________________________________________________________________________________________________________________
ABSTRACT
VaxTyVi® is a plain polysaccharide vaccine based on the capsular polysaccharide Vi (PsVi) from Salmonella Typhi, produced at Finlay Institute. Quantification of PsVi in VaxTyViTM, is a mandatory requirement for final product release and has being made by inhibition ELISA. Considering the disadvantages of this type of ELISA and that recently our group obtained a MAb against PsVi, the aim of this work, was to develop a sandwich ELISA to quantify PsVi in VaxTyViTM. M&M. Using MAb 4G3E11 as the capture and detection, a sandwich ELISA was developed. Limits of detection (LOD) and quantitation (LOQ) were calculated, as well as the inter- and intra-assay for each point of the standard curve. The PsVi content in six batches of VaxTyVi™ was also determined. Results. An eight-point calibration curve with a range from 50 to 0.39 ng/mL of PsVi was obtained with a coefficient of determination (R2) ≥0.99. The LOD and LOQ were 0.59 ng/mL and 1.25 ng/mL respectively, representing at least 1000 times more sensitive than inhibition ELISA. The inter-assay CV was 4–15%, and the intra-assay CVs varied between 3-8 % and 4–9% for intraplate and interplate respectively. The PsVi content in all VaxTyVi™ lots were found into the accepted range (±30%). In summary, the proposed sandwich shown to be specific, reproducible and highly sensitive. The assay shown to be suitable for use in the quantitation of PsVi in VaxTyViTM, with great potential to replace the inhibition ELISA, implanted in the Finlay Institute since 2006.
KEYWORDS: polysaccharide Vi quantification, sandwich ELISA, Typhoid fever, monoclonal antibodies
_______________________________________________________________________________________________________________________
RESUMEN
VaxTyVi® es una vacuna simple polisacárida basada en el polisacárido capsular Vi (PsVi) de Salmonella Typhi, producido en el Instituto Finlay. La cuantificación de PsVi en VaxTyViTM, es un requisito obligatorio para la liberación del producto final y se ha realizado mediante ELISA de inhibición. Teniendo en cuenta las desventajas de este tipo de ELISA y que recientemente nuestro grupo obtuvo un MAb contra PsVi, el objetivo de este trabajo, fue desarrollar un sándwich ELISA para cuantificar PsVi en VaxTyViTM. Utilizando el MAb 4G3E11 como captura y detección, se desarrolló un ELISA en sandwich. Se calcularon los límites de detección (LOD) y la cuantificación (LOQ), así como el inter e intra-ensayo para cada punto de la curva estándar. También se determinó el contenido de PsVi en seis lotes de VaxTyVi ™. Se obtuvo una curva de calibración de ocho puntos con un intervalo de 50 a 0,39 ng / ml de PsVi con un coeficiente de determinación (R2) ≥ 0,99. El LOD y LOQ fueron 0.59 ng / mL y 1.25 ng / mL respectivamente, lo que representa al menos 1000 veces más sensible que ELISA de inhibición. El CV inter-ensayo fue del 4-15%, y el CV intra-ensayo varió entre 3-8% y 4-9% para intraplaca e interplate, respectivamente. El contenido de PsVi en todos los lotes VaxTyVi ™ se encontró en el intervalo aceptado (± 30%). En resumen, el sandwich propuesto demostró ser específico, reproducible y altamente sensible. El ensayo demostró ser adecuado para su uso en la cuantificación de PsVi en VaxTyViTM, con gran potencial para reemplazar el ELISA de inhibición, implantado en el Instituto Finlay desde 2006.
PALABRAS CLAVE: cuantificación del polisacárido Vi, ELISA en sándwich, fiebre tifoidea, anticuerpos monoclonales
_______________________________________________________________________________________________________________________
INTRODUCTION
Typhoid fever, also known as enteric fever, is a potentially fatal multi systemic illness caused primarily by Salmonella Typhi and, to a lesser extent, related serovars paratyphi A, B, and C. Humans are the only source of these bacteria; no animal or environmental reservoirs have been identified. The microorganism is most often acquired through consumption of water or food that has been contaminated by feces of an acutely infected or convalescent person or a chronic, asymptomatic carrier. 1
Typhoid fever is still a health problem, an estimated 22 million cases and 200,000 related deaths occur worldwide each year. Its prevalence is higher in Asia, sub-Saharan Africa and some countries in South America, but 90% of fatal cases occur in Asia, specifically in India, Pakistan and Bangladesh. 2
In Cuba, there has been an increasing tendency to decrease morbidity and mortality from typhoid fever, describing two stages in the epidemiology of the disease: before and after 2002. Before 2002, an average of 178.4 cases reported per year, with a rate of 1.9 per 100 000 inhabitants. Since 2002, we observed a significant decrease in the incidence of the disease, coming from six cases per year to be no reports of the disease in 2013. 3 Figure 1.

Figure 1. Behavior of FT in Cuba. The average number of annual cases of FT shown in different periods: before 2002, from 2002 to 2012 and from 2013.
The change in the behavior of the disease since 2002, is related among other factors, with the introduction since 2001 of the Cuban typhoid vaccine, VaxTyViTM, in the National Immunization Program. VaxTyViTM is a plain polysaccharide (Ps) vaccine, based on the capsular polysaccharide Vi (PsVi) from S. Typhi, with proven efficacy and safety for 14 years of implementation and is produced at Finlay Institute. 4
According to international standards, quantification of PsVi in VaxTyViTM, is a mandatory requirement for final product release. Several techniques have been used for quantification of PsVi. Immunological assays (IA) such as rate nephelometry or solid phase immunoassay (ELISA) are within them. 5,6 In the case of ELISA, different types are described in the scientific literature to quantify antigens. 7 In Finlay Institute, Fajardo et al (2006) validated an inhibition ELISA to quantify the PsVi in VaxTyVi™ and until today, it is the method used by the quality control department for the final release of vaccine lots. 8 However, this type of ELISA has several disadvantages, among which we can mention: more laborious, consuming more steps including inhibition or neutralization, lower sensitivity and specificity (employs polyclonal serum), and difficult interpretation of the results, because antibody-antigen-enzyme complex bound is inversely related to the concentration of antigen present in the sample. 9 Sandwich ELISA nevertheless, is easier to perform, and in the last decade has been more used in the quantification of total and free Ps in polysaccharide vaccines like Haemophilus influenzae b Tetanus toxoid conjugate vaccines and multivalent antimeningococcal polysaccharide vaccines. 10,11
Recently our group obtained a MAb against PsVi coded as 4G3E11, with high purity, affinity and specificity confirmed. 12,13 The high specificity and affinity constant of the MAb 4G3E11 suggests its use as an analytical tool in enzyme immunoassays. The aim of the present work was to develop a sandwich ELISA to quantify the PsVi in VaxTyViTM and thus replace the ELISA inhibition assay, considering the disadvantages mentioned above.
Materials and methods
Reagents and buffers
The following buffers were used: coating buffer (15 mM Na2CO3, 35 mM NaHCO3, pH 9.6), PBS (140 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.8 mM KH2PO4, pH 7.4), blocking buffer (PBS, 3% non-fat dried milk), washing buffer (PBS, 0.05% Tween 20 (v/v), pH 7.4), and substrate buffer (35 mM citric acid, 67 mM Na2HPO4, 0.012% (w/v) H2O2, pH 5.0). All reagents were obtained from Sigma-Aldrich (USA).
MAbs
Murine MAb against PsVi were produced and characterized as described previously (12,13). Briefly, hybridomas were produced by fusion of SP2/O myeloma cell line with splenocytes of BALB/c mice that were immunized with three intraperitoneal doses (every 20 days) of PsVi conjugated to diphtheria toxoid (vaccine candidate from Finlay Institute). One hybridoma, coded as 4G3E11 were selected and produced in vivo grown as mouse ascites in the peritoneal cavity of pristane-primed BALB/c mice. The MAb were purified from the ascites fluid (AF) by affinity chromatography using Protein A (GE Healthcare, Germany) following manufacturer's instructions. As part of the characterization was determined: Productivity, Class and Subclass, Purity, Specificity and Affinity constant (Kaff) Table 1

Peroxidase labeling of MAbs
Purified MAb were conjugated in-house to horseradish peroxidase type VI (HRP) as described by Wilson (1978). Briefly, 8 mg of MAb was coupled to 4 mg of HRP and was purified by gel filtration chromatography. Conjugates containing 1% BSA, 0.05% thiomersal and 50% glycerol were stored in small aliquots at −20 °C.
Sandwich ELISA
Calibrators, quality controls and calibration curve construction
In the absence of International Standards, calibrators consisted of purified PsVi produced at Finlay Institute (Havana, Cuba) and used as internal controls (IC). PsVi concentration was quantitated using colorimetric Hestrin’s assay. The IC was supplied as lyophilized powders with 100 μg of PsVi. A two-fold serial dilution of IC was used to generate an eight-point calibration curve with a range from 50 to 0.39 ng/mL. Ascent software Version 2.6 (Thermo Labsystem Oy, USA) was used for plotting the optical density (OD492) against PsVi concentration and four-parameter logistic equation (4PL) was applied to obtain the function describing a sigmoid model. The quality control (QC) consisted of PsVi powders (Finlay Institute, Havana, Cuba) which were weighted, dissolved in PBS and adjusted to a final concentration of 5 ng/mL (w/v). Calibrators and QC were stored as single ready-to-use aliquots at −20 °C.
ELISA sandwich for PsVi quantitation
Samples
Six VaxTyViTM vaccine lots (9006, 0004, 402, 203, 304 y 405) released by quality control department, with known concentration of 50 µg/mL determined by ELISA inhibition, were evaluated. All VaxTyViTM lots were provided by Pharmaceutical Development Direction of Finlay Institute and were used at dilution of 1:10000, taking into account the standard curve range.
Procedure
Polystyrene microwell plates (Maxisorp, Nunc, USA) were coated with 10 μg/mL of MAb 4G3E11 anti PsVi, in coating buffer (100 μL/well). After overnight incubation at 4°C, the coated plates were washed three times and blocked with blocking buffer for 1 h at 37°C. The calibrators, QC and samples were added (100 μL/well) and incubated 2 h at 37°C. Subsequently, the wells were washed three times and peroxidase conjugated MAb 4G3E11 diluted 1:2500 in washing buffer containing 1% non-fat dried milk, were added to the wells (100 μL/well) and incubated for 1 h at room temperature. After four washes, 0.4 mg of OPD per mL of substrate buffer was added to the wells (100 μL/well) and following 30 min of incubation in the dark, the reaction was stopped with 1M H2SO4. OD was measured at 492 nm using a Microplate Reader (Multiskan Ex, Thermo LabSystem, USA). Unknown concentrations were calculated using Ascent Software Version 2.6 (Thermo LabSystem Oy, USA). The calibrator, QC and samples were run in triplicates unless otherwise stated. Three independent experiments were performed and results were expressed as mean of PsVi content. Optimal dilutions of MAb and HRP conjugated MAb were determined by standard checkerboard titrations according to the procedures described by Crowther and Walker (2009).
Limits of detection and quantitation
Lower limits of detection (LOD) and quantitation (LOQ) were determinated and defined as the mean of blank values (n = 20) plus either 3 or 10 standard deviation (SD) respectively.
Intra- and inter-assay precision
Intra-assay precision was determined by interplate and intraplate precision as calculated by the coefficient of variation (CV). The interplate precision was evaluated by measuring triplicates of standard curve on four distinct ELISA plates on the same day in parallel experiments. The intraplate precision was evaluated by measuring the standard curve in quadruplicate on a single ELISA plate. The inter-assay precision was determined as the CV on five separate occasions.
Results
Curve Construction
MAb 4G3E11 was used as the capture and a peroxidase conjugated MAb 4G3E11 was used for detection in a sandwich ELISA assay. An eight-point calibration curve with a range from 50 to 0.39 ng/mL of PsVi was obtained (Fig. 2). A four-parameter fit model was applied to obtain the function describing a sigmoid model. Coefficient of determination (R2) ≥0.99 was obtained.

Figure 2. Calibration curve obtained from sandwich ELISA using the Ascent™ Software Version 2.6 (Thermo LabSystem Oy, USA). A four-parameter logistic equation (4PL) was applied to obtain the function describing a sigmoid model. Error bars represent standard deviations of quadruplicate samples.
Sensitivity, Reproducibility and Repeatability
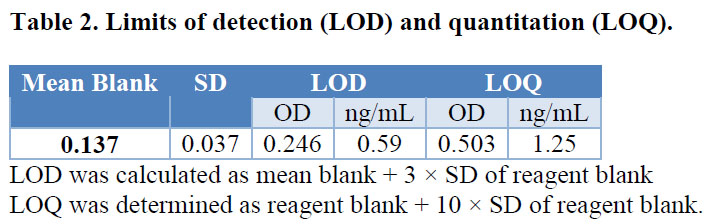
The LOD and LOQ were found to be 0.59 ng/mL and 1.25 ng/mL respectively and could be related with the Kaff value calculated for the MAb in the previous experiment. Table 2
The assay reproducibility and repeatability was determined by precision assay, calculating the inter and intra-assay expressed in CV% (Table 3).

As can be seen the proposed sandwich ELISA showed an inter-assay CVs of 4–15%, below 20% recommended for this type of assay. The intra-assay CVs varied between 3-8 % and 4–9% for the intraplate and interplate assay respectively, values that are below 10% recommended for intra-assay test in enzyme immunoassays. 14
PsVi quantitation in vaccines
The proposed sandwich ELISA was used to measure the PsVi content in six lots of VaxTyViTM (summarised in Table 4).

As shown in Table 5 all lots of VaxTyVi™ evaluated show concentrations of PsVi determined by the sandwich ELISA within the accepted range (± 30%) according to the product specifications (35 to 65 µg/mL).

All vaccines lots were found into the accepted range (±30%) of PsVi content according to the available European Pharmacopoeia guidelines and the CVs were <4% compared to the acceptance criterion of <15% CV for immunoassays. 15
DISCUSSION
Facile techniques to analyse and quantitate the Ps components of Ps-based vaccines are essential for the evaluation of antigen content and lot-to-lot consistency of manufacture, with the associated implications for efficacy and safety. Content of Ps is directly related to the product dose. 15,16 For PsVi quantitation, several methods are available and suitable for use. 17 The concentration of the PsVi in its fully O-acetylated, acid form in eluted fractions can be measured using Hestrin’s method 18 or another suitable method, such as Nuclear Magnetic Resonance (NMR). 19 The acridine orange staining method (20) and high performance anion exchange chromatography with pulsed amperometric detection (HPAEC-PAD) 21 have been reported to produce comparable results for PsVi in a range of 15–200 μg/ml. In the case of NMR and HPAEC-PAD, these two techniques have a high sensitivity (at least 10 times higher than Hestrin´s assay) but requires the use of expensive reagents and very sophisticated equipment, not accessible for all. 10
However, immunosorbent assays such as ELISAs, have been very useful in quantifying antigens in vaccines, especially for Ps antigens. 10,11 ELISAs have a variety of positive qualities that make them great attraction. 9 (Figure 3)

Figure 3. Scheme showing features of ELISA that make it advantageous for a wide range of applications. Four major advantages of ELISA are promoted, all of which add to the reasons that this form of ELISA has been, and will continue to be, successful. 9
In Finlay Institute since 2006, an inhibition ELISA was developed and standardized, to quantify PsVi in VaxTyViTM. 8 Although this ELISA has guaranteed over the years, the release of VaxTyViTM batches efficiently and consistently, it has several disadvantages compared to sandwich ELISA. The main disadvantages are related to the number of steps and the interpretation of results. 9
In this manuscript, we describe the use of a simple sandwich ELISA for the quantification of PsVi in VaxTyViTM. The proposed sandwich ELISA that use the MAb 4G3E11 for coating and the same MAb but conjugated to peroxidase, for detection, shown to be specific, reproducible and highly sensitive.
According to the sensitivity, related with the lowest amount of analyte which can be detected in a sample, in this paper was evaluated by calculating LOD. 22 In the proposed sandwich ELISA the LOD was 0.59 ng/mL, value representing at least 1000 fold more sensitive than the value reported for the inhibition ELISA (0.5 μg/mL). This big difference in the detection limit between both ELISAs, could be determined by the use of MAb in sandwich ELISA, regarding the use of polyclonal antibodies (serum) in the ELISA inhibition. Moreover, if we consider that the MAb 4G3E11 used, has a high specificity and affinity constant for the antigen (3.13 x 1011 M-1). 13
The reproducibility and repeatability of the method, was evaluated by assay precision, which takes into account the maximum dispersion of data obtained for a sample processed several times, on different days and in different conditions. The inter-assay and intra-assay CVs were calculated for each point of the standard curve (50–0.39 ng/mL) as described Reyes F. et al. 11 Reproducibility and repeatability were in accordance with international recommendations, which define the acceptance criterion of precision for standard curves with values below 10% and 20 % for intra and inter-assay precision respectively. 14 These CV values obtained for inter and intra-assay were lower than those reported by Fajardo et al (8) for inhibition ELISA (17-19.5% for inter assay and 16-19.8 % for intra assay). This result may be because, as mentioned above, in the inhibition ELISA, more steps are employed, including inhibition or neutralization step, compared with the sandwich ELISA. The use of larger number of steps, resulting in greater manipulation of the sample in the test by staff and therefore greater possibility of error, resulting in reproducibility.23
However, despite the high sensitivity and reproducibility of the proposed sandwich ELISA, was important to demonstrate the application of the assay, in quantify the amount of PsVi present in VaxTyViTM, and it was done. Six lots of VaxTyViTM were evaluated using the developed sandwich ELISA. All vaccines lots have an expected nominal label amount of 50 μg/mL of PsVi and all of concentrations obtained through sandwich ELISA, were within the accepted range (±30%) of PsVi according to the European Pharmacopoeia guidelines. 15 The CVs were <4% compared to the acceptance criterion of <15% CV for immunoassays. In ELISA inhibition, the CV obtained were between 4.7% and 12.3% for an average of 7.7%, higher than the sandwich ELISA but also within the acceptance criteria.
Therefore, these assays can be used successfully for the quantitation of PsVi in typhoid polysaccharide vaccines, even in conjugate vaccines. In addition, using the proposed sandwich ELISA and taking into account their high sensitivity, these assays could be a useful tool to test free PsVi in conjugated vaccines, prior separation of the unbound Ps from conjugated-Ps in such vaccines. Free polysaccharide must be tested to ensure that the amount present is within the limits agreed by the regulatory authorities based on lots shown to be clinically safe and efficacious. 17
In summary, the assay described here has been shown to be suitable for use in the quantitation of PsVi in VaxTyViTM, with great potential to replace the inhibition ELISA, implanted in the Finlay Institute since 2006.
Reference
1. Romero, R., (2007). Microbiología y parasitología humana. Salmonella. Ed. Médica Panamericana. ISBN 6077743542.
2. WHO. (2014). Typhoid Fever. Available on Internet at: http://www.who.int/topics/typhoid_fever.en/
3. MINSAP., (2013). Anuario Estadístico de Salud. Dirección Nacional de Registros Médicos y Estadísticas de Salud. Edición 42, Editorial MINSAP.
4. Riverón L., Cardoso D. y col. (2003). Vax-TyVi : Vacuna Cubana de Polisacárido Vi de Salmonella typhi. Biotecnología Aplicada; Vol.20, No.4
5. WHO Expert Committee on Biological Standardization. (2014). Guidelines on the quality, safety and efficacy of typhoid conjugate vaccines. WHO Technical Report Series No. 987, Sixty-fourth report. Annex 3
6. OMS. (2003) Manual de entrenamiento: concesión de licencias, liberación de lotes y disponibilidad de laboratorio. Vacunas y productos biológicos.
7. Voller A, Bidwell DR, Bartlett A. (1979). The enzyme-linked immunosorbent assay (ELISA). A guide with abstracts of microplate applications. Dynatech Europe, Borough House, Guernsey GB.
8. Fajardo E., Delgado I., Riverón L., y col. (2006). Validación de un ELISA tipo inhibición para cuantificar polisacárido Vi en la vacuna antitifoídica cubana VaxTyVi®. VacciMonitor, Año 15 No. 2
9. Crowther J. (2002). The ELISA Guidebook. METHODS IN MOLECULAR BIOLOGYTM. Huangzhiman 2002.12.18.
10. Saydam, M., Rigsby, P. and Mawas, F., (2014) A novel Enzyme-Linked Immuno-Sorbent Assay (ELISA) for the quantification of total and free polysaccharide in Haemophilus influenzae b-Tetanus toxoid conjugate vaccines in monovalent and combined vaccine formulations. Biologicals 42: 29-33
11. Reyes F. et al. (2014) Development of four sandwich ELISAs for quantitation of capsular polysaccharides from Neisseria meningitidIs serogroups A, C, W and Y in multivalent vaccines. Journal of Immunological Methods. 11834; No of Pages 5.
12. Amín N. y col. (2015). Obtención de un anticuerpo monoclonal murino que reconoce al polisacárido capsular Vi de Salmonella Typhi. VacciMonitor. 24(2):57-63
13. González E. y col. (2015). Producción, purificación y caracterización del AcM contra polisacárido capsular Vi de Salmonella Typhi y su aplicabilidad en ensayos de identidad. Revista BioProcesos. Vol.1, No.3.
14. Ochoa RF. (2013).Técnicas inmunoenzimáticas en el desarrollo clínico de vacunas. Finlay Ediciones;. p.3-13.
15. European Pharmacopoeia. (2002). Typhoid polysaccharide vaccine.4th Edition, Version 4.2, 07/2002:1160.
16. CECMED. (2000). Requisitos para la liberación de lotes de vacunas. Regulación No.19.
17. World Health Organization. (2013). Guidelines on the quality, safety and efficacy of typhoid conjugate vaccines. Adopted by the 64th meeting of the WHO Expert Committee on Biological Standardization, 21-25 October.
18. Hestrin S. (1949). The reaction of acetylcholine and other carboxylic acid derivatives with hydroxylamine, and its analytical application. Journal of Biological Chemistry, 180:249–261.
19. Lemercinier X. et al. (2000). Use and validation of an NMR test for the identity and O-acetyl content of the Salmonella typhi Vi capsular polysaccharide vaccine. Biologicals, 28:17–24.
20. Stone AL, Szu SC. (1988). Application of optical properties of the Vi capsular polysaccharide for quantitation of the Vi antigen in vaccines for typhoid fever. Journal of Clinical Microbiology, 26:719–725.
21. Micoli F et al. (2011). Vi-CRM 197 as a new conjugate vaccine against Salmonella Typhi. Vaccine, 29:712–720.
22. Armbruster DA. and Pry T. (2008). Limit of Blank, Limit of Detection and Limit of Quantitation. Clin Biochem Rev. Vol 29 Suppl (i) August, S49
23. Bartlett JW. and Frost C. (2008). Reliability, repeatability and reproducibility: analysis of measurement errors in continuous variables. Ultrasound Obstet Gynecol 2008; 31: 466–475.
Recibido:19 octubre 2016
Aprobado; 30 noviembre 2016
Elizabeth González Aznar, Fátima Reyes, Nevis Amín Blanco, Oscar Otero Alfaro, Maribel Cuello Pérez, Fidel Ramírez Bencomo and Reinaldo Acevedo Groguez
Filiation: Laboratory of Monoclonal Antibodies from the Biological Evaluation Department, Research Area, Institute of Vaccines Finlay. Havana, Cuba, A.P. 16017, Cod. 11600.
Author e-mail: [email protected]
