2022.07.02.19
Files > Volume 7 > Vol 7 No 2 2022
Biological activities of Ethanolic Extract Produced by Cucurbita pepo plant.
Dahlia Mohammed Ali Hasan1, *Butheina A. Hasoon2, Afnan I. Abdulwahab3, Kareem H.Jawad4
1,4 Department of Biomedical Engineering, Biomechanical Branch, University of Technology-Iraq
2,3Applied Science Department, University of Technology- Iraq.
* Correspondence: Butheina A. Hasoon . e-mail [email protected];07805283470.
Available from: http://dx.doi.org/10.21931/RB/2022.07.02.19
ABSTRACT
Antibacterial and antioxidant effects of alcoholic extract for the Cucurbita pepo plant were studied. Properties optical (UV-visible spectrophotometer ) and morphological surface(scanning electron microscopy ) detection of the alcoholic extract was done, and the antibacterial activity was investigated against pathogenic microbes (E.coli, Staphylococcus aureus). Results showed the highest effect for crud extract in the growth of E.coli by the zone of inhibition diameter reached (16.33±0.58 ), followed by Staph. Aureus by the diameter of the inhibition zone reached (12.33±2.30mm). The results indicate that the plant C. pepo can be considered a valuable source of effective antioxidant agents at a 0.8 µg/ml concentration.
Keywords. Cucurbita pepo plant: Alcoholic extract: Antioxidant: Antibacterial Activity
INTRODUCTION
Herbal medicine has long been utilized in India and China as a less expensive means of treating various health problems 1, and it is still the principal therapy for treating infections in several underdeveloped nations 2. When compared to synthetic drugs, herbal remedies are frequently less toxic and have fewer adverse effects. The World Health Organization has also advised that research be launched to identify and describe novel herbal remedies derived from historically recognized plants and create new effective therapeutic agents, particularly in places where modern, safe pharmaceuticals are lacking. For the treatment of long-term illnesses 3.
Aromatic herbs are commonly used as nutritional supplements 4, and they are treasured as a source of natural antioxidants 5. Because of the rising safety issues linked with the intake of synthetic antioxidants, it's more crucial than ever to seek cheaper and safer antioxidants from natural sources, mainly plants 6. C. pepo, a native squash plant, is one of the 15 species of Cucurbita genus in the Cucurbitaceae family 7. Gourds are utilized for various aesthetic and traditional purposes, while pepo vines and fruit are used as cattle feed 8,9. C. pepo is a superb protein, carbohydrate, mineral, and fat supplement 10. The cucurbits family, often known as cucurbits, is a vast collection of plants that may be cultivated in warmer climates worldwide and yield popular food crop plants. Some of these kinds are squash, gourd, watermelon, and gourd are some of these varieties. One of the first known cultivated species is the peppo squash. Fruits that are not fully ripe are eaten as vegetables. Sweet, ripe fruit is utilized in sweets and roasted or boiled beverages 11. This study aims to determine some biological activities of the C. pepo plant.
MATERIALS AND METHODS
Extract Preparation
The extraction was done by Soxhlet through 75% ethanol; then the solvent was removed using a rotary evaporator, then transferred to the oven to dry and stored at 4 ̊C until used12.
Phytochemical Screening of C. pepo extract:
The crude extract screening for the determination of phytoconstituents 13.
Characterization C. pepo extract
UV-Visible , and SEM
UV-Vis absorption spectra were employed for the optical test of C. pepo extract at various preparation conditions within range ( 365-540 nm ); the solution was measured by UV-Vis double beam astrophotometers SP-3000 plus (CE OPTMA TOKYO). All spectra were measured at room temperature. The size and shape of C. pepo extract were passed out by (SEM), and were applied in the University of technology-Iraq.
Antioxidant activity
Antioxidant activity of C. pepo was studied by DPPH assay with modified according to 13, ten µl of the sample was mixed up with 490 µl of DPPH and finally added 500 ethanol. The equation formula examined the optical density (O.D.) was examined by 517 nm by the equation formula13,16.
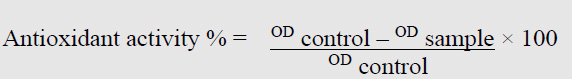
OD= Optical density
Preparation of bacterial isolates
Microbial isolates were taken from the Biotechnology department at the University of technology. The microbes were grown overnight at 37 ̊C on nutrient broth to prepare the suspensions cell to 0.5 McFarland standards (1 × 105 CFU/mL)16.
Antimicrobial activity
Isolates were obtained from the Biotechnology Branch/Applied Science Department at the University of Technology. The diffusion agar method was used to detect antimicrobial activity. The inhibition zone was measured around each well, compared with the control, and conducted in triplicate 12,13,16.
RESULTS
The UV-visible spectroscopy was used to check the formation alcohol extract of C. pepo. The surface of the alcohol extract band at 390 nm (Fig. 1).
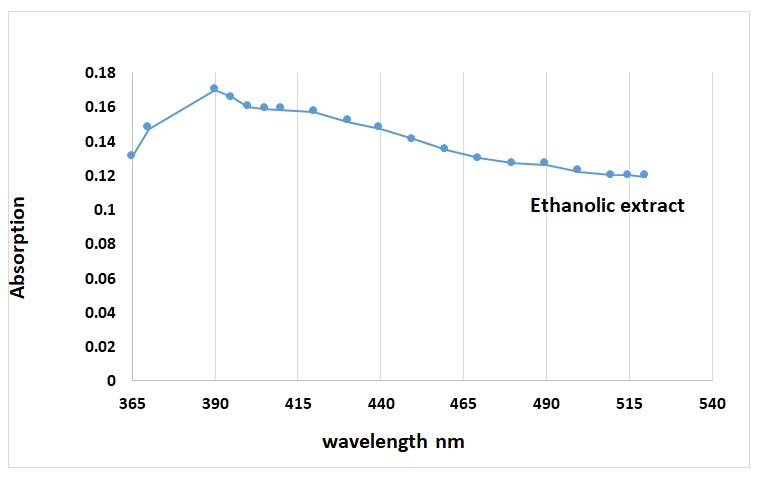
Figure 1. UV-Vis of the ethanolic extract by C. pepo plant.
SEM assay
SEM technique was employed to visualize the shape and size of the ethanolic extract by C. pepo plant shown in figure 2.

Figure 2. SEM of the ethanolic extract by C. pepo plant.
Phytochemical Screening of extract
The result of the Phytochemical Screening of plant extract ware dark green color suggests the presence of phenolics and tannin, respectively. Yellow to colorless (flavonoids), volatile oil by yellow precipitate, reddish-brown alkaloids, flavonoids by yellow precipitate is indicated by the formation of a precipitate, such as yellow precipitate, the presence of saponins is indicated by the persistence of frothing. While Glucosides, Steroids, and Terpenoids were absent in Ethanolic extract (Table 1).

Table 1 Phytochemical Screening of C. pepo plant.
Antioxidant activity of C. pepo plant
The DPPH was found to be proportional to the increase in concentration. The free radicals were given 56.50%, 62.42%.,66.12%,74.33% by the concentration of 20,40,60 and 80 μg/ml respectively in figure.3.

Figure 3. DPPH radical scavenging of C. pepo plant
Antibacterial activity
C. pepo extract effect was studied against two types of pathogenic microbes. The effects of the ethanolic extract varied on pathogenic microbes' growth. The highest effect observed on the growth of E. coli by a zone of inhibition reached (16±0.13mm), followed by S. aureus (15±0.58 mm) (Figure 4, and Table 2 ).

Figure 4. Antibacterial activity of ethanolic extract toward. E.coli, S. aureus.

Table 2. The effect of ethanolic extract against some pathogenic.
DISCUSSION
C. pepo extract exhibited antibacterial action related to Gram-negative and Gram-positive bacteria13. The activity of C. pepo extract against bacteria was credited to phytochemical materials like Flavonoids, phenolics, alkaloids, and tannins which affect the cell wall bacteria 14. Antibacterial activity differed depending on the cell wall structure of G +ve and G-ve bacteria15,18, which leads to the destruction of bacteria 16,18. In-creased phenolic elements, such as flavonoids, phenolic diterpenes, and phenolic acids, increased DPPH radical scavenging activity. Phenols, Flavonoids, and Alkaloids isolated from leaves of plants decreased the oxidant action 17.
CONCLUSIONS
C. pepo plants have wide phytochemical compounded and clinical investigations. Scientific studies have shown most of the claims of traditional medicines through has antibacterial activity against G+ve and G-ve bacteria, the ability of the extract to inhibitor 75%, and antioxidants.
Funding: "This research was funded by Dahlia Mohammed Ali Hasan, Butheina A. Hasoon, Afnan I. Abdulwahab, Kareem H.Jawad.
Informed Consent Statement:
"Written informed consent has been obtained from the biotechnology department and biomedical engineering at the University of technology
Data Availability Statement: The results might be achieved using an evaluation software or assessment table, available at www.diagnostics.sk/idmicro.
Acknowledgments include personal support "self-financing" (e.g., materials used for experiments).
Conflicts of Interest: None
REFERENCES
1. 1. Yadav, M.; Jain, S.; Tomar, R.; Prasad, G.S. and Yadav, H. Medicinal and biological potential of pumpkin : an updated review Nutrition Research Reviews Nutrition Research Reviews. 184–190 ,2017. doi:10.1017/S0954422410000107.
2. 2. Kennedy, D.A.; Lupattelli, A.; Koren, G.; Nordeng, H. Safety classification of herbal medicines used in pregnancy in a multinational study. BMC Complem Altern M .16, 102.(2016).
3. 3. Caili, F. U.; Huan, S. H. and Quanhong, L. A Review on Pharmacological Activities and Utilization Technologies of Pumpkin. 73–80 2006. doi:10.1007/s11130-006-0016-6.
4. 4.Jančić R, Stošić D, Mimica-Dukić M, Lakušić B. Aromatic plants of Serbia. Belgrade, Gornji Milanovac: NIP Dečje Novine. pp 296.1995.
5. 5. Tiwari, B.K. ; Valdramidis, V.P. ; Donnel, C.P.; Muthukumarappan, K; Bourke, P.; Cullen P.J. Application of natural antimicrobials for food preservation J Agric Food Chem, 57 , pp. 5987-6000, 2009.
6. 6. Zheng,W; S.Y. Wang Antioxidant activity and phenolic compounds in selected herbs J Agric Food Chem, 49 , pp. 5165-5170, 2001 .
7. 7. Bermejo, J.E. and León, J. eds. Neglected crops: 1492 from a different perspective. FAO. 1994.
8. 8. Kirtikar, K.R. and Basu, B.D. Indian medicinal plants. Indian Med Plants, 1985.
9. 9. Caili, F.U.; Huan, S. and Quanhong, L.I. A review on pharmacological activities and utilization technologies of pumpkin. PLANT FOOD HUM NUTR , 61(2): 70-77.2006.
10. 10. Yadav M .; Jain S.; Tomar R.; Prasad G. and Yadav H. Medicinal and biological potential of pumpkin. Nut. Res. Rev. Adebayo, O.R., Farombi, A.G., Oyekanmi, A.M. Proximate, Mineral, 23(2): 184–190 ,2010.
11. 11. Martha, R.; and Gutierrez, P. Medicinal chemistry Review of Cucurbita pepo ( Pumpkin ) its Phytochemistry and Pharmacology. 6, 12–21 ,2016.
12. 12. Abdulwahab, A.I.; Hasoon, B.A.; Maeah, R. and F. Al-azaw K.Preparation of Mentha Crispata Extract and Detection of its Biological Application. Sys Rev Pharm ,12(1):1151-1155, 2021.
13. 13. Hasoon, B.A.; Abdulwahab, A.I., Maeah, R. and F. Al-azaw K.. Preparation and Characterization of Silver Nanoparticle by Cordia myxa Extract and their Study Anticancer, Antioxidant, Antibacterial Activity . Indian Journal of Forensic Medicine & Toxicology, Vol. 15, No. 3.2021.
14. 14.Sugnay R. S.; Priya K. S. and Roxy B., phytochemical screening and antibacterial activity from Nerium oleander and evaluate their plant mediated nanoparticles synthesis, Inte. Res. J. Ph, 3(5), 285-288, 2012.
15. 15.Qi, Y.; Chin, K; Malekian, F.; Berhane, M. and Gager, J. Biological characteristics, nutritional and medicinal value of roselle, Hibiscus sabdariffa. Circular-urban forestry natural resources and environment, 604, 1-2,2005.
16. 16. Hasson, B.A.; Mahdi, L. H. and Essa, R.H. Evidence of Antioxidant Activity of Novel L-Glutaminase Purified from L. Gasseri BRLHM. Journal of Applied Sciences and Nanotechnology, Vol 4. 2021. DOI: 10.53293/jasn.2021.3969.1064.
17. 17. Maeah RK, Hasoon B.A, Abd-Alwahab AI, Al- Azawi KF, Hameedi WB. Biosynthesis of silver nanoparticles using Hibiscus sabdariffa and their biological application. EurAsian Journal of Biosciences. 2020 Sep 22;14(2):3377-83.
18. 18. Jawad K.H.; Saleh TH, Hasson A. B; Preparation of Aluminum Oxide Nanoparticles by Laser Ablation and a Study of Their Applications as Antibacterial and Wounds Healing Agent. Nano Biomed. Eng. 2019;11(3):313-9.
Received: 29 December 2021 / Accepted: 15 February 2022 / Published:15 May 2022
Citation: Dahlia Mohammed Ali Hasan1; Butheina A. Hasoon; Afnan I. Abdulwahab; Kareem H.Jawad. Biological activities of Ethanolic Extract Produced by Cucurbita pepo plant. Revis Bionatura 2022;7(2) 19. http://dx.doi.org/10.21931/RB/2022.07.02.19
