2022.07.03.13
Files > Volume 7 > Vol 7 No 3 2022
Molecular characterization of endophytic fungi from pine (Pinus oocarpa) in Honduras
Bryan Ortiz1, Lourdes Enríquez1, Karla Mejía2, Yensi Yanez2, Yuliana Sorto3, Sayda Guzman3 Kateryn Aguilar1, Gustavo Fontecha1, *
1 Microbiology Research Institute; Universidad Nacional Autónoma de Honduras; J1 building. Ciudad Universitaria, Tegucigalpa 11101, Honduras. [email protected] (B.O); [email protected] (L.E); [email protected] (K.A); [email protected] (G.F).
2 Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre (ICF), Colonia Brisas de Olancho, Comayagüela, Honduras. [email protected] (K.M); [email protected] (Y.Y).
3 Escuela Agrícola Panamericana (EAP). Departamento de Ciencia y Producción Agropecuaria, Laboratorio de control biológico, San Antonio de Oriente, Francisco Morazán, Honduras. [email protected] (Y.S); [email protected] (S.G).
* Correspondence: [email protected] Tel.: (+50433035342;) (BO).
Available from: http://dx.doi.org/10.21931/RB/2022.07.03.13
ABSTRACT
Pine is one of the important coniferous genera in America. In Honduras, Pinus oocarpa is the most critical conifer species since it plays an essential role for the country's economy and represents a symbol of national identity. Endophytic fungi are defined as microorganisms that reside in the internal tissues of plants without causing any obvious immediate negative effects and are an integral part of their associated microbial communities. This study aimed to isolate and characterize the endophytic fungi present in the pines of Honduras through the amplification and sequencing of the ribosomal RNA gene's internal transcribed spacer (ITS). A total of 7 pines from the department of El Paraíso in eastern Honduras were analyzed. A total of 14 fungi grouped into 6 genera and 7 species were isolated: Fusarium lateritium, F. pseudocircinatum, Pestalotiopsis pini, P. microspora, Xylaria grammica, Trichoderma atroviride, y Nigrospora oryzae. To our knowledge, this is the first report of endophytic fungal species in pines in Honduras. Although some endophytic fungi may be mutualistic or saprophytic, the present study shows the presence of several genera of endophytic fungi that have been reported as pine pathogens. The presence of these fungi in the pines of Honduras represents a potential threat to the health of the forest. Further research is needed to increase knowledge about the importance of these fungi and the potential impact they could have on pine forests in Honduras.
Keywords: Endophytic fungi, Pinus oocarpa, Honduras
INTRODUCTION
Pinaceae is the most important of the eight families of conifers. Within this family are grouped 11 genera distributed in boreal, arid, subalpine, temperate and tropical forest ecosystems1, 2. The Pinus genus is one of the most important within this family and includes more than 100 species distributed mainly in the forests of the countries of the northern hemisphere in Europe, Asia, North Africa, North America, and Central America. "Pines", as they are commonly called, have high ecological importance since they are the predominant tree species in many of the countries of these geographical regions3. Also, pine trees represent a fundamental pillar in the economies of several countries, and its by-products contribute significantly to the Gross Domestic Product (GDP), mainly in low- and middle-income countries that export the wood as raw material4, 5.
Endophytic fungi are microorganisms that reside in the internal tissue of plants without causing any apparent immediate adverse effects and that, together with other microorganisms, are an integral part of the microbial community of the plant6, 7. The association between fungi and plants represents a universal and cryptic phenomenon in nature6. Most of these microorganisms are transmitted horizontally through the production of conidia or spores that spread to adjacent uninfected plants using physical or mechanical agents6. Most of the endophytic fungi belong to the Ascomycota phylum; however, some genera belonging to the Glomeromycota, Basidiomycota and Zygomycota phyla have also been reported6, 8. Endophytic fungi are associated with plants as saprophytes, mutualists, or parasites7, 9, 10, although these fungi are generally found in host tissue as mutualists9. Factors such as stress, physical changes and the level of maturation of the plants are some of the causes that could be involved in the development of infections by this microorganisms7.
Endophytic fungi have been recognized as possible disease-causing agents in conifers, mainly in pines 11-13. Honduras has an estimated forest cover of 6,314,815 hectares, of which 1,951,978 hectares (30.91%) are pine forests14 including seven species and five varieties: P. oocarpa var. oocarpa and var. trifoliata, P. maximinoi, P. caribaea var. hondurensis, Pinus ayacahuite var. ayacahuite, P. hartwegii, P. pseudostrobus var. pseudostrobus, and P. tecunumanii15. In Honduras, pine is the most critical conifer species, and Pinus oocarpa has been selected as a national tree for its relevant economic role and scenic importance. Given the pathogenic potential that some genera of endophytic fungi could represent and due to the economic and cultural importance of pine trees in Honduras, this study aimed to characterize endophytic fungi isolated from pine using molecular methods.
MATERIALS AND METHODS
A total of 7 pine trees (Pinus oocarpa Schiede ex Schltdl) from the department of El Paraíso in Honduras were analysed. The trees showed a change in the color of the needles from their apex to the base, exudation of resin at the base of the fascicles, and vascular damage (Figure 1). Endophytic fungi were isolated using the superficial sterilization technique, previously described by Stone et al.7. Briefly, 4 samples of 1 cm of pine needles were randomly selected from each tree. The outside of the needles was sterilized with 4% sodium hypochlorite solution for 3 min, then rinsed three times with sterile distilled water. Once disinfected, the needles were cultured independently on PDA (Potato Dextrose Agar) and 10% tartaric acid. The Petri dishes were incubated at 25ºC and observed daily for 7 days to detect the appearance of colonies (Figure 1), then all the isolates were purified using the hyphal tip or single spore methods. DNA extraction was performed by inoculating the mycelium of each strain in 3 ml of GYEP broth (2% glucose, 0.3% yeast extract, 1% peptone)16. All cultures were incubated for two days at 26ºC. Subsequently, the mycelium mats were recovered and subjected to a protocol based on organic solvents. Briefly, the mycelium was resuspended in 500 µl of lysis buffer composed of 10 mM Tris, pH 8; 1 mM EDTA, pH 8; and 100 mM NaCl. This suspension was heated to 100ºC for 1 min in a water bath and then shaken three times in a Disruptor Gene® system (Scientific Industries) with 0.1 mm glass beads for 2 min with rest intervals of 1 min. 400 μL of phenol-chloroform-isoamyl alcohol was added (25:24:1), vortex mixed and centrifuged at 13,000 rpm for 15 min. The supernatant was recovered, and a volume of chloroform-isoamyl alcohol (24:1) was added, mixed with vortex, and centrifuged at 13,000 rpm for 5 min. The aqueous phase was transferred to a new vial and precipitated with a volume of absolute cold ethanol and 1/10 of the volume with sodium acetate (3M, pH 5.2), mixed by inversion and centrifuged at 13,000 rpm. The supernatant was removed, and the sediment was subsequently washed with 70% ethanol. The dried pellet was resuspended in 50 μL of nuclease-free water. The DNA concentration was calculated with a NanoDrop® spectrophotometer (Thermo Fisher Scientific Inc.), diluted to a final concentration of 40 ng/μL and stored to −20ºC until further use.
Molecular identification of all isolates was performed by amplification and sequencing of the internal transcribed spacer (ITS) of the nuclear ribosomal DNA gene. The ribosomal ITS region is recognized as a universal marker for the identification of fungi17. Polymorphisms in both length and sequence, the availability of databases to compare sequences, universal primers and a high rate of successful amplification among all fungal lineages are some of the reasons why this marker has been recognized as a good outlet for the identification of endophytic fungi8, 9, 17, 18.
PCR amplifications were carried out under the following conditions in a 50 μL volume: 25 μL of PCR Master Mix (Promega Corp. Madison, WI, USA), 1 μL of 10 μM ITS1 and ITS4 primers 5’-TCCGTAGGTGAACCTGCGG-3’/5’-TCCTCCGCTTATTGATATGC3', and 1 μL of DNA (40 ng/ μL). Reactions were carried out with an initial denaturation step at 95ºC for 5 min, 37 cycles of 95ºC for 30 s, 55ºC for 30 s, and 72ºC for 30 s, with a final extension at 72ºC for 10 min. Amplicons were visualized in 1.5% agarose gel electrophoresis with ethidium bromide. The amplified products were sequenced on both flanks with their respective primers according to Psomagen company protocols (https://lims.psomagen.com/). The quality of the sequences was analyzed in the software with the Geneious®9.1.7 software and queried against international databases contained in NCBI to confirm the identity of the sequences. The sequences were compared with sequences deposited in GenBank, registering the result with the highest percentage of similarity as the most likely identification of each isolate. The sequences were deposited in GenBank, and accession numbers were assigned for 12 isolates.

Figure 1. (A) Pine needle lesions and resin exudation at the base of the fascicles; (B) Endophytic fungal growth from pine needles.
RESULTS AND DISCUSSION
A total of 14 monoaxenic isolates were obtained from the 7 trees analyzed. All samples were successfully amplified and sequenced. The size of the amplicons ranged from 500 to 700 bp. The sequencing results yielded 6 genera and 7 different species. The endophytic fungi identified were Fusarium lateritium, F. pseudocircinatum, Pestalotiopsis pini, P. microspora, Xylaria grammica, Trichoderma atroviride, and Nigrospora oryzae. The most frequently identified species was P. pini, while Trichoderma atroviride and Xylaria grammica were the least frequent (Table 1). All the fungal isolates in this study belong to the Ascomycota phylum, which agrees with what has been reported in the literature regarding the classification of endophytic fungi in plants6, 8, 19. However, the isolates were grouped under the Sordariomycetes class, and this finding contrasts with what was previously reported in Germany and Spain, where the Dothideomycetes class was the most frequent in Pinus silvestris19, 20. Something similar happened with the endophytes described in Pinus nigra subsp.
On the other hand, endophytic fungi of the Arthoniomycetes class were the most frequent in Pinus silvestris from Sweden24, and the Leotiomycetes class was the most commonly found in the United States and South Korea in Pinus taeda, P. leiophylla and P. densiflora respectively25-27. The Sordariomycetes class was the second in frequency in Spain, South Korea and the USA19, 26, 27, while in a study carried out in Germany, fungi of the class Sordariomycetes were found in 31% of all isolates20. Moreover, according to what was reported by Bullington et al. in the USA and Taudiere et al. in Croatia this class was the least frequent in both studies21, 22. The high diversity of microorganisms that pine species can harbor in different geographic regions is evident, and multiple factors could influence the prevalence and distribution of fungal communities in coniferous species. Further studies are needed to clarify the main determinants of the prevalence of various classes of fungi in this group of trees.
Of 14 isolates, 6 (42.9%) belonged to two species of the genus Pestaloptiopsis: P. pini, (n = 4) and P. microspora (n = 2). Fungi grouped in this genus are generally considered secondary pathogens that can be responsible for a wide variety of plant diseases including cankers, dieback, leaf spots, needle blight, tip blight, grey blight, and severe chlorosis, among others13. The presence of Pestalotiopsis spp. in pines has been reported in Iraq, Spain, Portugal, China, USA, Slovakia and some other European countries 13, 28-32, but as far as we know this is the first report of two species of the genus Pestalotiopsis in Honduras.
Similarly, Fusarium lateritium (n = 2) and F. pseudocircinatum (n = 1) were identified in this study. Fusarium spp. it is among the main genera of endophytic fungi. This genus includes important plant pathogens that affect both forest and agricultural species. Although the presence of F. lateritium and F. pseudocircinatum has been reported in pine trees33, there is little information in the literature on these two species that cause pine infection. Romon et al. suggested that F. lateritium was a potential agent for the biological control of infections by phytopathogens, mainly F. circinatum34. It is important to highlight that, although the ITS regions are considered a reliable universal marker for identifying Fusarium, the combination of at least two loci could be more informative and conclusive for the discrimination between species, which is a limitation in this study.
On the other hand, two isolates of the genus Nigrospora, one Xylaria grammica and one Trichoderma atroviride were identified. These fungi have also been identified as endophytic microorganisms of pine trees in the western part of the Himalayas, Spain, and China19, 35. To our knowledge, this is the first report of these three genera of endophytic fungi isolated from Pinus oocarpa in Honduras.
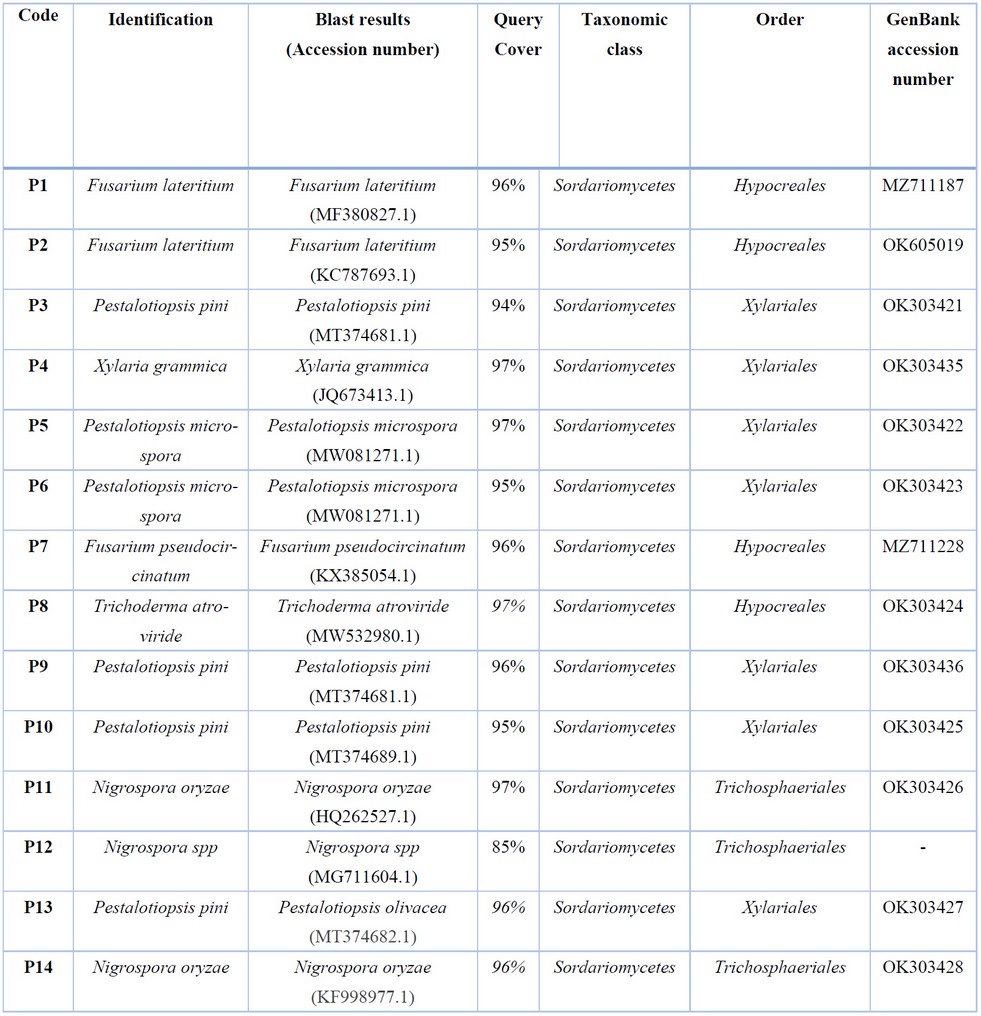
Table 1. Identified species of endophytic fungi and accession numbers assigned by GenBank.
CONCLUSIONS
This is the first report of endophytic pine fungi isolated in Honduras. The presence of these fungi in the pines of Honduras represents a potential threat to the health of the pine forests. The information of these fungi in the country is limited and more studies are needed to increase our knowledge about the presence of these microorganisms and the potential impact they could have on the forest.
Author Contributions:
B.O., L.E., and G.F., conceptualized the study; K.M., Y.Y., Y.S., S.G., K.A., performed the mycological diagnosis; B.O., K.A., performed molecular experiments; B.O., K.A., Y.S., S.G., L.E., and G.F., organized and cured the data; writing and original draft preparation, B.O., and G.F.; All the authors contributed with writing, review, and editing the manuscript; supervision, project administration, and funding acquisition, B.O., K.M., Y.Y., L.E., All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding
Informed Consent Statement: Any research article describing a study involving humans should contain this statement. Please add "Informed consent was obtained from all subjects involved in the study." OR "Patient consent was waived due to REASON (please provide a detailed justification)." OR "Not applicable." for studies not involving humans. You might also choose to exclude this statement if the study did not involve humans.
Written informed consent for publication must be obtained from participating patients who can be identified (including by the patients themselves). Please state "Written informed consent has been obtained from the patient(s) to publish this paper" if applicable.
Data Availability Statement: In this section, please provide details regarding where data supporting reported results can be found, including links to publicly archived datasets analyzed or generated during the study. Please refer to suggested Data Availability Statements in section "Bionatura Research Data Policies" at https://www.revistabionatura.com/policies.html. You might choose to exclude this statement if the study did not report any data.
Acknowledgements: The authors thank the Staff of the Forest Health and Sanitation Unit of El Paraíso department for the collection of the samples and their technical support to achieve the goals of the project. The project was carried out with financing provided by Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre de Honduras (ICF).
Conflicts of Interest: The authors declare no conflict of interest.
REFERENCES
1. Nkongolo K, Mehes-Smith M. Karyotype evolution in the Pinaceae: implication with molecular phylogeny. Genome. 2012;55(10):735-53.
2. Gernandt DS, López GG, García SO, Liston A. Phylogeny and classification of Pinus. Taxon. 2005;54(1):29-42.
3. Nobis MP, Traiser C, Roth-Nebelsick A. Latitudinal variation in morphological traits of the genus Pinus and its relation to environmental and phylogenetic signals. Plant Ecology & Diversity. 2012;5(1):1-11.
4. Küçüker D, Baskent E. State of stone pine (Pinus pinea) forests in Turkey and their economic importance for rural development. Options Méditerranéennes: Série A Séminaires Méditerranéens; Carrasquinho, I, Correia, AC, Mutke, S, Eds. 2017:111-7.
5. Moctezuma López G, Flores A. Economic importance of pine (Pinus spp.) as a natural resource in Mexico. Revista mexicana de ciencias forestales. 2020;11(60):161-85.
6. Tadych M, White JF, Moselio S. Endophytic microbes. Encyclopedia of Microbiology ed M Schaechter. 2009:431-42.
7. Stone JK, Polishook JD, White JF. Endophytic fungi. Biodiversity of Fungi Elsevier Academic Press, Burlington. 2004:241-70.
8. Vasundhara M, Reddy MS, Kumar A. Secondary metabolites from endophytic fungi and their biological activities. New and future developments in microbial biotechnology and bioengineering. 2019:237-58.
9. Zakaria L, Aziz WNW. Molecular identification of endophytic fungi from banana leaves (Musa spp.). Tropical life sciences research. 2018;29(2):201.
10. Sour V, SarayutPhonpho K. Antifungal activities of endophytic fungi isolated from orchids against Colletotrichumgloeosporioidescaused anthracnose in orchids. Journal of Agricultural Technology. 2015;11(8):1949-61.
11. Pirttilä AM, Pospiech H, Laukkanen H, Myllylä R, Hohtola A. Two endophytic fungi in different tissues of Scots pine buds (Pinus sylvestris L.). Microbial Ecology. 2003;45(1):53-62.
12. Peršoh D. Factors shaping community structure of endophytic fungi–evidence from the Pinus-Viscum-system. Fungal Diversity. 2013;60(1):55-69.
13. Silva AC, Diogo E, Henriques J, Ramos AP, Sandoval-Denis M, Crous PW, et al. Pestalotiopsis pini sp. nov., an Emerging Pathogen on Stone Pine (Pinus pinea L.). Forests. 2020;11(8):805.
14. Instituto de Conservación Forestal I. Anuario Estadistico Forestal de Honduras, 3era edición. Centro de Información y Patrimonio Forestal, Unidad de Estadisticas Forestales. Available at: https://icf.gob.hn/?portfolio=cipf-2. Last access September, 27,2021. . 2020.
15. Padilla. EG. Estado de la diversidad biológica de los árboles y bosques de Honduras. Documentos de Trabajo: Recursos Genéticos Forestales. FGR/51S Servicio de Desarrollo de Recursos Forestales, Dirección de Recursos Forestales, FAO, Roma. (Inédito). Available at: https://www.fao.org/3/j0607s/j0607s01.htm#TopOfPage Last access November,01,2021. . 2003.
16. Tang CM, Cohen J, Holden DW. An Aspergillus fumigatus alkaline protease mutant constructed by gene disruption is deficient in extracellular elastase activity. Molecular Microbiology. 1992;6(12):1663-71.
17. Schoch CL, Seifert KA, Huhndorf S, Robert V, Spouge JL, Levesque CA, et al. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proceedings of the National Academy of Sciences. 2012;109(16):6241.
18. Vasić T, Jevremović D, Krnjaja V, Leposavić A, Anđelković S, Živković S, et al. Morphological description and molecular detection of Pestalotiopsis sp on hazelnut in Serbia. Spanish Journal of Agricultural Research. 2017;15(3):e10SC02.
19. Sanz-Ros AV, Müller MM, San Martín R, Diez JJ. Fungal endophytic communities on twigs of fast and slow growing Scots pine (Pinus sylvestris L.) in northern Spain. Fungal biology. 2015;119(10):870-83.
20. Bußkamp J, Langer GJ, Langer EJ. Sphaeropsis sapinea and fungal endophyte diversity in twigs of Scots pine (Pinus sylvestris) in Germany. Mycological Progress. 2020;19(9):985-99.
21. Taudiere A, Bellanger J-M, Carcaillet C, Hugot L, Kjellberg F, Lecanda A, et al. Diversity of foliar endophytic ascomycetes in the endemic Corsican pine forests. Fungal ecology. 2018;36:128-40.
22. Bullington LS, Larkin BG. Using direct amplification and next-generation sequencing technology to explore foliar endophyte communities in experimentally inoculated western white pines. Fungal Ecology. 2015;17:170-8.
23. Oono R, Lefèvre E, Simha A, Lutzoni F. A comparison of the community diversity of foliar fungal endophytes between seedling and adult loblolly pines (Pinus taeda). Fungal biology. 2015;119(10):917-28.
24. Millberg H, Boberg J, Stenlid J. Changes in fungal community of Scots pine (Pinus sylvestris) needles along a latitudinal gradient in Sweden. Fungal Ecology. 2015;17:126-39.
25. Oono R, Rasmussen A, Lefèvre E. Distance decay relationships in foliar fungal endophytes are driven by rare taxa. Environmental Microbiology. 2017;19(7):2794-805.
26. U'Ren JM, Riddle JM, Monacell JT, Carbone I, Miadlikowska J, Arnold AE. Tissue storage and primer selection influence pyrosequencing‐based inferences of diversity and community composition of endolichenic and endophytic fungi. Molecular Ecology Resources. 2014;14(5):1032-48.
27. Eo J-K, Park H, Eom A-H. Diversity of Endophytic Fungi Isolated from Pinus densiflora and Juniperus rigida Distributed in Mt. Baekryeonsan and Mt. Johangsan, Korea. The Korean Journal of Mycology. 2018;46(4):437-46.
28. Maharachchikumbura SS, Hyde KD, Groenewald JZ, Xu J, Crous PW. Pestalotiopsis revisited. Studies in Mycology. 2014;79:121-86.
29. Botella L, Diez JJ. Phylogenic diversity of fungal endophytes in Spanish stands of Pinus halepensis. Fungal Diversity. 2011;47(1):9-18.
30. Cleary M, Oskay F, Doğmuş HT, Lehtijärvi A, Woodward S, Vettraino AM. Cryptic risks to forest biosecurity associated with the global movement of commercial seed. Forests. 2019;10(5):459.
31. Chen J, Hao X, Liu X, Ma L. First Report of Pestalotiopsis neglecta Causing Black Spot Needle Blight of Pinus sylvestris var. mongolica in China. Plant Disease. 2020;104(5):1545-.
32. Qi M, Xie C-X, Chen Q-W, Yu Z-D. Pestalotiopsis trachicarpicola, a novel pathogen causes twig blight of Pinus bungeana (Pinaceae: Pinoideae) in China. Antonie van Leeuwenhoek. 2021;114(1):1-9.
33. Nirenberg HI, O'Donnell K. New Fusarium species and combinations within the Gibberella fujikuroi species complex. Mycologia. 1998;90(3):434-58.
34. Romón P, Troya M, Fernández de Gamarra M, Eguzkitza A, Iturrondobeitia J, Goldarazena A. Fungal communities associated with pitch canker disease of Pinus radiata caused by Fusarium circinatum in northern Spain: association with insects and pathogen-saprophyte antagonistic interactions. Canadian Journal of Plant Pathology. 2008;30(2):241-53.
35. Guo LD, Huang GR, Yu W, ZHENG WH. Molecular identification of white morphotype strains of endophytic fungi from Pinus tabulaeformis. Mycological Research. 2003;107(6):680-8.
Received: 20 March 2022 / Accepted: 25 july 2022 / Published:15 Agoust 2022
Citation: Ortiz B, Enríquez L, Mejía K, Yanez Y, Sorto Y, Guzman S, Aguilar K, Fontecha G. Molecular characterization of endophytic fungi from pine (Pinus oocarpa) in Honduras. Revis Bionatura 2022;7(3) 13. http://dx.doi.org/10.21931/RB/2022.07.03.13
