2023.08.03.75
Files > Volume 8 > Vol 8 No 3 2023
Atherogenic Index of Plasma is a Novel Biomarker Associated with Obesity in the Adult Males
1 The University of Al-Ameed, Karbala PO Box 198, Iraq; [email protected]
2 Kufa University, Najaf, Iraq; [email protected]
3 Najaf Health Department; [email protected]
4 Kufa University, Najaf, Iraq; [email protected]
* Correspondence: [email protected]
Available from: http://dx.doi.org/10.21931/RB/2023.08.03.75
The atherogenic index of plasma (AIP) is a vital parameter for dyslipidemia and its associated diseases and assessing cardiac risk. Several anthropometric parameters have been used to reflect obesity-associated risk, but body mass index (BMI) and waist/ hip (W/H) ratio are the most common. Our study aimed to evaluate the correlation between AIP and BMI, waist/ hip W/H ratio and its advantage in predicting dyslipidemia among obese males. The study includes 869 healthy Iraqi males, 415 obese and 454 non-obese males. The mean ± standard deviation (SD) body mass index was 33.00 ± 2.76 kg/m2 for the obese males and 22.49 ± 1.21kg/m2 for the non-obese males. AIP, BMI, W/H and lipid levels were estimated. The study demonstrated that AIP level was significantly correlated with both BMI, Pearson Correlation 0.295** and W/H ratio, Pearson Correlation 0.297**. The study revealed that AIP was significantly and positively associated with BMI and W/H ratio among the study population.
Keywords: Obesity; Lipid; Atherogenic index of plasma; Males
INTRODUCTION
Obesity is considered a significant public health and economic trouble, leading to disturbing serum lipoprotein concentration; these changes are considered substantial risk factors for atherosclerotic cardiovascular events1. In each reign in the world, the prevalence of obesity increases constantly; thus, the correlated economic and medical costs, mortality and morbidity are also predicted to be elevated. Most of these complexities are associated with co-morbid cases involving type 2 diabetes mellitus, dyslipidemia, heart disease and hypertension2. Obese individuals are usually observed to have disorders in lipid metabolism. About 60-70% of obese subjects are dyslipidemic3. Abnormalities of the lipid levels in obese individuals involve increased serum LDL-c, TG and VLDL-c levels4. The AIP is described as a logarithmic ratio of plasma levels of triglyceride (TG) to high-density lipoprotein cholesterol (HDL-c) [log10 (TG/HDL-c)] and is potentially related to cardiovascular events and metabolic syndrome5. Increased plasma lipid levels make obese individuals more susceptible to atherosclerotic vascular disease and thrombosis.AIP can act as an adjunct to the individual lipid profile and is considered the best indicator to determine the fractionated esterification rate of HDL-c. In addition, it is more valuable than routine lipid parameters. It can also be used as a diagnostic value when the other atherogenic risk parameters appear normal6. The AIP measurement estimates the indicator of the zone of atherogenic risk ( AIP= -0.3-0.1 associated with low CV risk, 0.1-0.24 with medium and above 0.24 with high CV risk)7. A recent study has indicated that AIP reflects the correlation between atherogenic and protective lipoprotein and stands out as a potent predictor of coronary heart disease and atherosclerosis, which might comprehensively reflect the balance between the atherogenic and anti-atherogenic parameters8.
A case-control study involving 869 healthy Iraqi males, 415 obese and 454 non-obese males, was performed at the Department of Biochemistry at the College of Medicine / University of Kufa; this study was established between the beginning of October 2020 and the end of November 2021. The sample size was estimated using Epi Info (version 3). Subjects chosen for the study were categorized as non-obese and obese as stated by the WHO (World Health Organization), obese with BMI ≥ 30 and non-obese persons with BMI between 18.5 to 24.9. BMI was calculated using the formula BMI = weight in kg/height in m2. The atherogenic index of plasma (AIP) was calculated as log10 (TG/HDL-C)9. The criteria for inclusion of the study subjects (obese and non-obese) were healthy males with no evidence of any chronic illness, including hepatic, renal or thyroid. At the same time, the exclusion criteria involved individuals with a history of heart failure, stroke or diabetes, smoking, and lipid-lowering agents like fibrates or statins. Women were not included to eliminate the variation in the results that may arise from the differences in this parameter and to remove the effect of any confounding factors related to lipid profile.
Blood samples were collected from all participants after at least 12 hours of fasting. Lipids analysis was identified on fasting non-obese and obese males. Serum total cholesterol (TC), TG and HDL-c were carried by enzymatic colorimetric methods (Biolabo/France), while low-density lipoprotein (LDL-c) levels were measured by Friedewald formula10.
Data analysis was summarised by mean ± SD and investigated using IBM Statistical Package for Social Science Program SPSS version 24. The correlation between various variables was estimated using the Pearson correlation coefficient. A p-value less than 0.05 was considered significant.
For comparison between obese and non-obese males, significant differences were noticed for TG, TC, LDL-c, HDL-c, BMI and AIP with P-value < 0.001. The mean ± SD serum levels of TG, TC, and LDL-c were significantly increased among obese compared with the non-obese males, except for HDL-C levels were markedly decreased in obese compared with non-obese males. Regarding the W/H ratio, the obese group was significantly associated with a high W/H ratio and similarly exhibited an extremely positive association with increased BMI and AIP (P-value < 0.0001) compared with the non-obese group (Table 1).
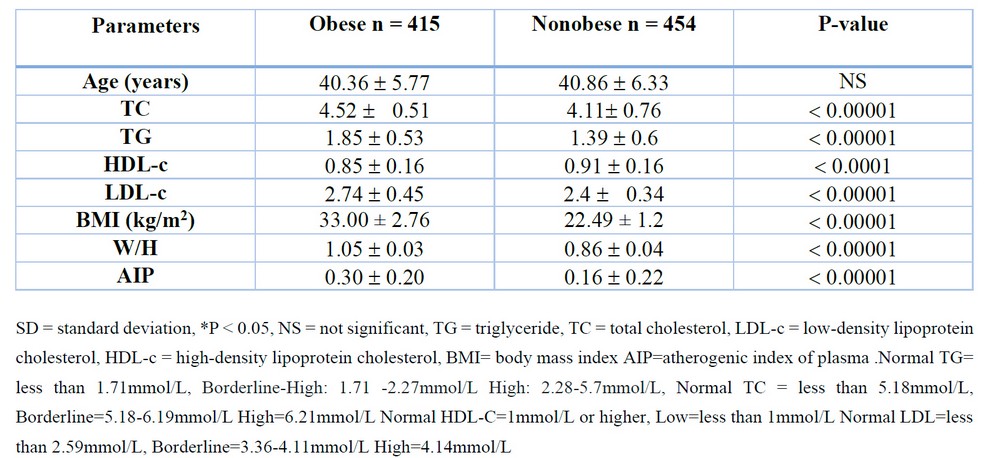
Table 1. Anthropometric and biochemical parameters of the study population
For the analysis of the coloration between BMI and W/H Ratio with AIP parameter among 869 participant males, the examination of data revealed that AIP was significantly correlated with BMI and W/H ratio, Pearson Correlation (0.295** 0.297**) respectively and P value (< 0.001) (Figure 1, Table 2).
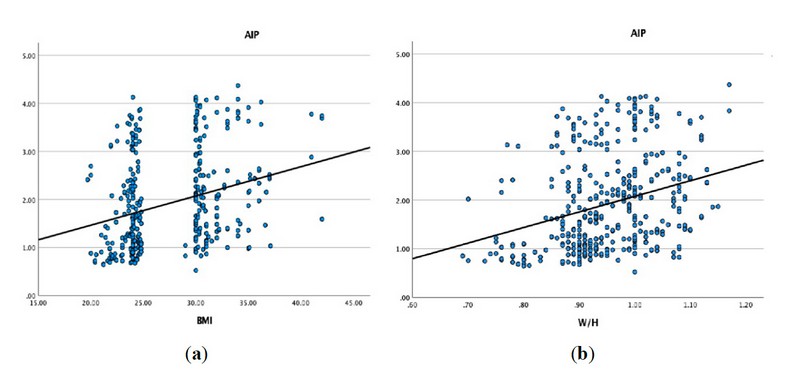
Figure 1. Correlation between BMI and W/H Ratio AIP parameter among the study population(a) AIP was significantly correlated with BMI; (b) AIP was associated considerably with W/H ratio.
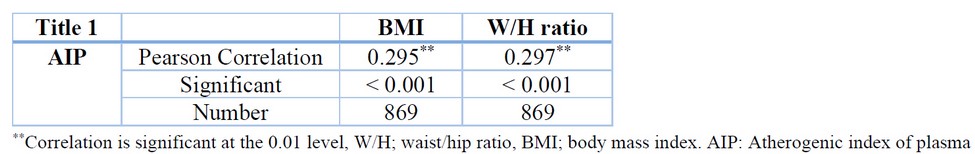
Table 2. Pearson Correlation between BMI and W/H Ratio with AIP parameter in the study population
Our study estimated the association between AIP and obesity in adult males. All obese males had increased lipid levels compared with non-obese males Table 1. These findings are in agreement with several former studies11-13. The association between hyperlipidemia and obesity is well-determined, and this association contributes to the risk of cardiovascular disease14. It was demonstrated that within obese males, Hypertriglyceridemia is considered a significant cause of dyslipidemia since it will result from impaired clearance of the TG-rich lipoprotein in chylomicron and VLDL-c. Hence, the VLDL-c and LDL delipidation processes are also inadequate, leading to increased levels of remnant lipoproteins and LDL-c15. Obesity is described mainly by fat accumulation in the subcutaneous parts of the body16. It has been demonstrated that accumulation of excessive body fat may increase the progression of various cardiac risk factors, including dyslipidemia, insulin resistance and hypertension, which are suggested as the primary causes leading to obesity-induced coronary artery diseases17. Body mass index BMI and W/H ratio as health risk parameter has several limitations because BMI does not accurately describe various components of body constituents, and waist-to-hip ratio W/H is an excessively used anthropometric parameter of central obesity. Therefore, AIP is considered a novel index that has been used to quantify lipid levels, and it was revealed that individuals with higher levels of AIP tended to be at an increased risk of dyslipidemia18. In our study, the data showed that AIP was strongly positively correlated with BMI and W/H ratio, Pearson Correlation (0.295** 0.297**) respectively and P value (< 0.001) Table 2. It was reported that integrating two parameters (TG, HDL-c) to generate AIP is a better biomarker for obesity19. AIP was considered an indicator of plasma atherosclerosis and correlated with important parameters like LDL-c and LDL-c20.
Interestingly, Yildiz G et al.21 revealed that AIP was significantly associated with intima-media thicknesses. A large-scale case-control study in China also reported that AIP was associated considerably with cardiovascular disease22. It was demonstrated that the W/H ratio correlates strongly with cardiovascular disease risk parameters (TG, HDL-c, hypertension and diabetes) when compared with BMI23. Further, Nevill AM et al.24 supported these findings and suggested the superiority of the W/H ratio to BMI. These findings exhibited that the W/H ratio and AIP are parameters related to the metabolism of lipids, and an elevation in the W/H ratio indicates an alteration in fat deposition in the abdomen. In contrast, an increase in AIP is referred to as dyslipidemia.
This study was a noteworthy strength of a large-scale case-control study. Our study reached the exciting finding that the atherogenic index of plasma was significantly and positively correlated with BMI and W/H ratio among obese and non-obese males, and it is easily estimated.
Author Contributions: Conceptualization, RA and TA; methodology, RA and FA; software, AHFA; validation, RA, AH and TA; formal analysis, RA and FA; investigation, AH; resources, RA; data curation, RA; writing—original draft preparation, RA; writing—review and editing, AH; visualization, TA; supervision, TA; project administration, RA All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding
Institutional Review Board Statement: The medical ethical committee in the Kufa Medical College approved this study. The study was conducted according to the guidelines of the Declaration of Helsinki.
Informed Consent Statement: Informed consent was obtained from all subjects involved in the study
Conflicts of Interest: The authors declare no conflict of interest.
1. Stadler JT, Marsche G. Obesity-related changes in high-density lipoprotein metabolism and function—International Journal of Molecular Sciences. 2020 Nov 26;21(23):8985.
2. Okunogbe A, Nugent R, Spencer G, Ralston J, Wilding J. Economic impacts of overweight and obesity: current and future estimates for eight countries. BMJ global health. 2021 Oct 1;6(10):e006351.
3. Feingold KR. Obesity and dyslipidemia. Endotext. 2020 Nov 2.
4. Pluta W, Dudzińska W, Lubkowska A. Metabolic Obesity in People with Normal Body Weight (MONW)—Review of Diagnostic Criteria. International Journal of Environmental Research and Public Health. 2022 Jan;19(2):624.
5. Li YW, Kao TW, Chang PK, Chen WL, Wu LW. Atherogenic index of plasma as predictors for metabolic syndrome, hypertension and diabetes mellitus in Taiwan citizens: a 9-year longitudinal study. Scientific reports. 2021 May 10;11(1):1-8.
6. Fan B, Zhang T, Li S, Yan Y, Fan L, Bazzano L, He J, Chen W. Differential Roles of Life-course Cumulative Burden of Cardiovascular Risk Factors in Arterial Stiffness and Thickness. Canadian Journal of Cardiology. 2022 Mar 18.
7. Academy of Medicine of Malaysia, Malaysian Clinical Practice Guidelines on Management of Type II Diabetes Mellitus, Academy of Medicine of Malaysia, Kuala Lumpur, Malaysia,5th edition, 2015.
8. Alaminos-Torres A, Martínez-Álvarez JR, López-Ejeda N, Marrodán-Serrano MD. Atherogenic Risk, Anthropometry, Diet and Physical Activity in a Sample of Spanish Commercial Airline Pilots. International Journal of Environmental Research and Public Health. 2022 Mar 31;19(7):4128.
9. Nwagha UI, Ikekpeazu EJ, Ejezie FE, Neboh EE, Maduka I. Atherogenic index of plasma as useful predictor of cardiovascular risk among postmenopausal women in Enugu, Nigeria. African health sciences. 2010;10(3).
10. Cicero AF, Fogacci F, Patrono D, Mancini R, Ramazzotti E, Borghi C, D'Addato S, Bove M, Piani F, Giovannini M, Grandi E. Application of the Sampson equation to estimate LDL-C in children: Comparison with LDL direct measurement and Friedewald equation in the BLIP study. Nutrition, Metabolism and Cardiovascular Diseases. 2021 Jun 7;31(6):1911-5.
11. Ikeda K, Morizane S, Akagi T, Hiramatsu-Asano S, Tachibana K, Yahagi A, Iseki M, Kaneto H, Wada J, Ishihara K, Morita Y. Obesity and Dyslipidemia Synergistically Exacerbate Psoriatic Skin Inflammation. International journal of molecular sciences. 2022 Apr 13;23(8):4312.
12. Luo J, Wang JK, Song BL. Lowering LDL cholesterol: from mechanisms to therapies. Life Metabolism. 2022 May 20.
13. Norwitz NG, Soto-Mota A, Kaplan B, Ludwig DS, Budoff M, Kontush A, Feldman D. The Lipid Energy Model: Reimagining Lipoprotein Function in the Context of Carbohydrate-Restricted Diets. Metabolites. 2022 May;12(5):460.
14. Paquette M, Bernard S, Paré G, Baass A. Dysbetalipoproteinemia: Differentiating Multifactorial Remnant Cholesterol Disease From Genetic ApoE Deficiency. The Journal of Clinical Endocrinology & Metabolism. 2022 Feb;107(2):538-48.
15. Al-Samawi RI, Smaism MF. Association of the lipoprotein lipase and Apolipoprotein C-II gene polymorphisms with risk of dyslipidemia in smokers and non-smokers male. Indian Heart Journal. 2022 Jan 1;74(1):45-50.
16. Piché ME, Tchernof A, Després JP. Obesity phenotypes, diabetes, and cardiovascular diseases. Circulation research. 2020;126(11):1477-500
17. Bays HE. Evaluation and Practical Management of Increased Visceral Fat: Should Cardiologists Lose Sleep Over It?. Journal of the American College of Cardiology. 2022 Apr 5;79(13):1266-9.
18. Onuchukwu IC, Etim EU, Chimdike OG, Oguzie BC, Emuebie H, Orji KN, Egoh LC, Ofojama EL, Okonkwo GI, Timothy AO, Igwe CC. Review on Atherogenic Index of Plasma Lipids and Dyslipidemia. Journal of Drug Delivery and Therapeutics. 2022 Aug 15;12(4-S):214-22.
19. Zhu X, Yu L, Zhou H, Ma Q, Zhou X, Lei T, Hu J, Xu W, Yi N, Lei S. Atherogenic index of plasma is a novel and better biomarker associated with obesity: a population-based cross-sectional study in China. Lipids in Health and Disease. 2018 Dec;17(1):1-6.
20. Huang H, Yu X, Li L, Shi G, Li F, Xiao J, Yun Z, Cai G. Atherogenic index of plasma is related to coronary atherosclerotic disease in elderly individuals: a cross-sectional study. Lipids in health and disease. 2021 Dec;20(1):1-9.
21. Yildiz G, Duman A, Aydin H, Yilmaz A, Hür E, Mağden K, Çetin G, Candan F. Evaluation of assoc ation between atherogenic index of plasma and intima‐media thickness of the carotid artery for subclinic atherosclerosis in patients on maintenance hemodialysis. Hemodialysis International. 2013 Jul;17(3):397-405.
22. Moussavi Javardi MS, Madani Z, Movahedi A, Karandish M, Abbasi B. The correlation between dietary fat quality indices and lipid profile with Atherogenic index of plasma in obese and non-obese volunteers: A cross-sectional descriptive-analytic case-control study. Lipids in Health and Disease. 2020 Dec;19(1):1-9.
23. Tsai HS, Tseng WK, Yin WH, Lin FJ, Hsuan CF, Wu YW, Huang LC, Lin TH, Chang KC, Li YH, Yeh HI. The correlation between waist-hip ratio and achieving therapeutic lipid goals in Taiwan. Acta Cardiologica Sinica. 2019 Nov;35(6):605.
24. Nevill AM, Stewart AD, Olds T, Duncan MJ. A new waist-to-height ratio predicts abdominal adiposity in adults. Research in Sports Medicine. 2020 Jan 2;28(1):15-26.
Received: 25 June 2023/ Accepted: 26 August 2023 / Published:15 September 2023
Citation: Al-Samawi R, Al-Kashwan T A, Alsailawi F A, Algenabi A H A. Atherogenic Index of Plasma is a Novel Biomarker Associated with Obesity in the Adult Males. Revis Bionatura 2023;8 (3) 75. http://dx.doi.org/10.21931/RB/2023.08.03.75
