CS 2019.02.01.13
Files > Conference Series > 2019 > Humboldt Kolleg 2019
Bionatura Conference Series Vol 2. No 1. 2019
“Breaking Paradigms: Towards a Multi-, Inter- and Transdisciplinary Science” In commemoration of the 250th Anniversary of Alexander von Humboldt
Microalgae species as potential inhibitors of the bioactivity of Vibrio parahaemolyticus in shrimps
Freddy Figueroa, Bryan Aldaz
available in: http://dx.doi.org/10.21931/RB/CS/2019.02.01.13
ABSTRACT
Vibrio parahaemolyticus is a Gram-negative motile bacteria belonging to Vibrionaceae family causing cytotoxicity in cultured cells and enterotoxicity in animal models. Similarly, this bacterium is a major foodborne pathogen that causes life-threatening diseases in human, including gastroenteritis and septicemia, via consumption of contaminated seafood. Based on the increasing interest in developing novel environmental friendly solutions for the inhibition of the bioactivity of V. parahaemolyticus, microalgae species have emerged as potential candidates due to the microbiota associated with microalgae cultures, oxygen radicals produced during the photosynthetic process and the production of secondary metabolites, useful for antibacterial properties. In the present review, conventional treatments concerning V. parahaemolyticus bioactivity have been presented. However, in terms of effectiveness, numerous studies explaining the use of microalgae species for antibacterial activity against V. parahaemolyticus have been detailed suggesting that microalgae-based treatment are considerably better. Finally, we presented a proposed microalgae-based solution that can act as an inhibitor of V. parahaemolyticus bioactivity and therefore can be applied in the aquaculture industrial field to improve productivity and reduce costs.
Keyword: Vibrio parahaemolyticus, microalgae, antibacterial activity, shrimps, productivity
INTRODUCTION
Vibriosis is considered as an infection caused by Vibrio bacteria concerning mainly marine species including fishes and shrimps 1-3. However, this is not a holistic definition because the pathology and involved strains are not the same for the larvae and fattening and therefore, must be analyzed individually 4. Vibriosis, as bacterial pathology, has been the cause of mortality in shrimp culture in producer countries around the world, including Ecuador, affecting larviculture in its fattening phase in the shrimp farm 5, 6. Normally, vibriosis appears due to the abrupt changes in the environmental conditions leading to an increase in the mate bacterial rate overcoming the withstanding amount by organisms such as shrimps and they are Gram-negative bacteria belonging to Vibrionaceae family 7. The main pathogen species reported in fish and shrimps are V. alginolyticus, V. anguillarum, V. campbellii, V. carchariae, V. damsella, V. fischeri, V. harveyi, V. logei, V. mediterranei, V. nigripulchritudo, V. ordalii, V. orientalis, V. parahaemolyticus, V. pelagicus, V. penaeicida, V. splendidus and V. vulnificus 8-10.
It is well known that species associated with vibriosis family have long-term pathologic side effects involving humans causing gastroenteritis, septicemia and wound infections and aquatic animals with economic and environmental impact 11. Similarly, vibriosis affects marine species such as fishes, crabs, and shrimps. Therefore, there is a necessity of search for novel environmental friendly solutions for vibriosis. Previous studies have presented many solutions for enhancing the vibrio resistance of affected species or protecting them against the vibriosis. Sung et al. have proven that beta-glucan dispensed in concentrations of 0.5 mg/mL and 1 mg/mL in tiger shrimp can significantly increase their disease resistance 12. Sotomayor and Balcazar have demonstrated that a mixture of probiotic strains inhibits Vibrio sp. pathogens proliferation thus reducing their presence in aquaculture systems 13. Ma et al. have developed a novel polyvalent attenuated live bacterial vaccine to prevent and cure vibriosis of fish produced by the action of V. anguillarum and V. alginolyticus 14. Nevertheless, there is an evident attraction about using derived-algae species such as microalgae for the prevention of the vibriosis diseases and enhancing of vibrio resistant in marine species, specifically shrimps 15.
Marine algae are one of the most ancient components of the plant kingdom having rich biodiversity potential 16. Due to its unique properties, it is helpful against helminthic diseases and cancer. Algae has been of particular interest for the development of bioactive compounds for treatment/remedies for numerous types of bacterial diseases including tuberculosis, multidrug-resistant bacteria, a viral infection like HIV, Herpes viruses and vibriosis 17, 18. Several studies have demonstrated that algae species have a potential effect as an antimicrobial agent for the treatment of bacterial infections in aquaculture 19. Silva et al. have tested many solvents (i.e. ethanol, methanol, chloroform) extracts of different macroalgae species for the inhibition of various Vibrio strains 20. Besides, microalgae have also depicted bactericidal effects against vibriosis, according to Widowati et al. study 21. Further studies performed by Lauritano demonstrated that microalgae have anti-infective properties 22.
In the present review, we hypothesized that the biodiverse source of worldwide microalgae species (i.e. Chlamydomonas reinhardtii, Chlorella vulgaris, Klebsormidium flaccidum, Cosmarium impressulum, Kirchneriella lunaris, and Spirulina platensis) can be potential inhibitors of the bioactivity of V. parahaemolyticus in shrimps. Furthermore, we presented the T3SS1 effectors’ mechanism, three conventional treatments, the microalgae-based treatment, and a proposed novel, environmental friendly microalgae-based solution for the inhibition of Vibrio strains inside marine organism useful for the aquaculture industry.
Vibriosis parahaemolyticus: T3SS1 effectors mechanism
Activities of T3SS1 effectors in cultured intestinal epithelial cells are illustrated in figure 1. An uncharacterized protein has the gene VPA0450 that hydrolyzes phosphatidylinositide (4, 5)-biphosphate (PI(4,5)P2) to D5 phosphate (PI4P) and breaks the bond between the membrane and the actin cytoskeleton, leading to membrane blebbing. Vop Q inhibits the MAPK pathway by acetylating MKK, and Vop S is an adenylyltransferase involved in virulence by mediating the addition of adenosine 5'-monophosphate (AMP) to specific threonine residue of host Rho GTPases RhoA, Rac and Cdc42 23.

Figure 1. Schematic representation of the T3SS1 effector events cascade present in cultured intestinal epithelial cells triggered by V. parahaemolyticus.
The resulting AMPylation prevents the interaction of Rho GTPases with downstream effectors, thus inhibiting actin assembly in infected cells as well as inhibits Rho by AMPylation leading to cells rounding and phagocytes intrusion. In addition, Vop S binds to PIP2 in the membrane, thereby promoting the refolding of the T3SS effectors 23. Additionally, Table 1 presents in more detail every single T3SS1 effectors with their biological activity, their involved genes as well as their effects on host cells.
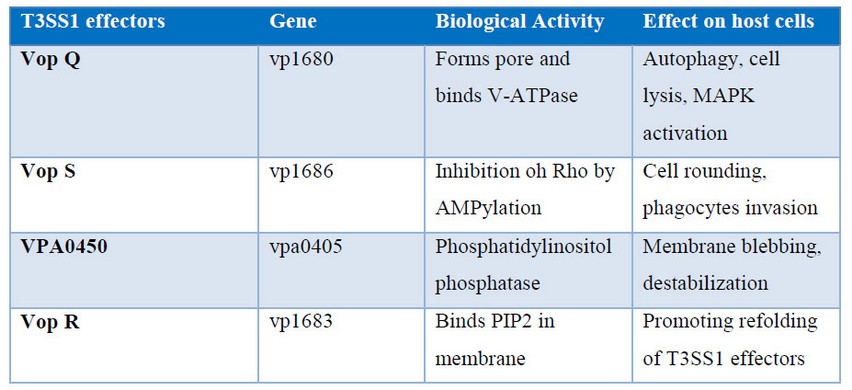
Table 1. Biological effects of T3SS1 effectors 23.
Vibriosis parahaemolyticus: Current treatments
Nowadays, traditional treatments used in aquaculture are based on the use of low-amperage electricity, acidic electrolyzed water treatment, X-ray and the use of the well-known antimicrobial compounds 24-27. Continuedly, in this review, three different conventional methods to treat and avoid growth and proliferation of V. parahaemolyticus are presented.
Acidic electrolyzed water treatment. According to Wang et al., the acidic electrolyzed water (AEW) treatment notably decreased the growth rate of V. parahaemolyticus extending its lag time. In addition, AEW treatment demonstrated the accelerated death of the bacteria at temperatures ≤ 10 °C. They successfully demonstrated the potential growth inhibition or death acceleration of V. parahaemolyticus on seafood, including shrimps. 25
Effect of X-ray on the inherent microflora in ready-to-eat shrimp. This method is based on the use of X-ray to decrease the growth rate of V. parahaemolyticus inoculated in ready-to-eat shrimps. The inoculated ready-to-eat shrimps were put into sterilized bags and treated with 0.1, 0.2, 0.3, 0.5, 0.75, 1.0, 2.0 3.0 and 4.0 KGy X-ray at ambient conditions of temperature. The obtained results depicted a reduction of more than six colonies formation units (CFU) using 3.0 KGy X-ray. However, the quality was not assessed leading to possible unsafety inoculated ready-to-eat shrimps. 26
Antimicrobial compounds. Former studies reveal that the use of commercial antibiotics for treating diseases produced inadmissible side effects. Immanuel et al. proved that the herbal diet of encapsulated powder of antimicrobial compounds extracted from terrestrial plants such as Ricinus communis, Phyllanthus niruri, Leucus aspera, Manihot esculenta, and seaweeds Ulva lactuca and Sargassum wightii gave better results than the use of commercial antibiotics leading to disease-free shrimp with high production 27. Their results of this investigation suggest that a diet based on antimicrobial compounds reduce the risk of contaminated shrimps. Shrimps raised in V. parahaemolyticus non-inoculated water and fed with conventional food exhibited the best survival (86.10%), specific growth rate (SGR, 2.87%) and the least bacterial load in muscle and hepatopancreas tissues. In contrast, shrimps raised in V. parahaemolyticus inoculated medium and fed with conventional food showed the lowest survival (24.44%), specific growth rate (1.11%) and the most bacterial load in muscle and hepatopancreas tissues. The shrimps fed with herbal diet noticeable stimulated the survival (43.32 – 58.88%), specific growth rate (1.46 – 2.15%) and lowered V. parahaemolyticus load in muscle and hepatopancreas tissues in the culture system 27.
V. parahaemolyticus: Microalgae-based treatment
Numerous studies have demonstrated that various microalgae species present antimicrobial properties for different pathogenic bacteria including Vibrio bacterial strains 28-33. Their anti-infective or bacteria-resistant properties can be attributable to the production of secondary metabolites, which are accumulated in the growth medium at the end of the exponential and the stationary phase of growth 31, 32. Their bacterial effects can also be related to (i) the microbiota associated with microalgae cultures and (ii) oxygen radicals produced during the photosynthetic process 33. Therefore, microalgae have emerged as a suitable environmental friendly solution for treating vibriosis concerning shrimps in the aquaculture industry.
Transgenic microalgae. Studies performed by Li et al. suggest that microalga Nannochloropsis oculata was useful for the development of transgenic microalgae presenting a bactericidal defense against V. parahaemolyticus. They cultured N. oculata in order to provide an organism (e.g. fish, shrimps) protection against bacterial pathogen infection. Researchers constructed an algae-codon-optimized bovine lactoferricin (LFB) fused with a red fluorescent protein (DsRed) driven by a heat-inducible promoter, which is a heat shock protein 70A promoter combined with a ribulose-1,5-bisphosphate carboxylase/oxygenase small subunit 20 promoters from Chlamydomonas reinhardtii, which is a microalga. They conducted an electroporation process for gene transfer. Once the electroporation is completed, two stable transgenic lines were generated, each one expressing a stable transgene inheritance for many months. Positive detection of the mRNA transcript and the protein of LFB-DsRed confirmed the transgene inheritance of cell lines. Their results proved that organisms fed with the LFB-containing transgenic microalgae would have a bactericidal defense against V. parahaemolyticus 28.
Estuarine microalgae. Norichika et al. carried out a study to demonstrate the attachment of V. parahaemolyticus strains to different microalgae strains. They prepared six distinct strains on modified Bristol agar plates, five corresponding to Navicula and one to Chlorella microalgae species. They used strains of thermostable direct hemolysin (TDH)-producing and TDH-non-producing V. parahaemolyticus. Once both parts are fully prepared, they mixed microalgae and live bacteria at a specific density and then incubated at 25°C for 6 hours. To estimate the viable counts present in supernatant, they utilized modified Bromothymol-blue-lactose agar plates supplemented with nalidixic acid or rifampicin. Therefore, a reduction of viable counts compared with the control value may be due to the attachment of V. parahaemolyticus strains to microalgae. Results obtained from this experiment clearly depicted a diminution of viable counts in Navicula-derived estuarine strains, thus confirming the main hypothesis of study 29.
Antibacterial activity in microalgae cultures. Experiments conducted by Kokou et al. have tested and successfully shown that five cultures of microalgae (Chlorella minutissima, Tetraselmis chui, Nannochloropsis sp., Arthrospira platensis, and Isochrysis sp.) present the ability to inhibit the growth of Vibrio bacterial strains. Furthermore, they evaluated the influence of light on the antibacterial activity of the microalgae. Cultures of these five microalgae species with no culturable bacteria were used in a first series of in vivo experiments. Subsequently, incubation of bacteria in a culture of microalgae (co-culture) in order to measure the ability of these microalgae species to inhibit the growth bacterial strains including Vibrio parahaemolyticus, Vibrio anguillarum, Vibrio splendidus, Vibrio scophthalmi, V. alginolyticus, and V. lentus. In the second series of experiments, they tested the influence of light by preparing two different environments, one with light conditions and the other in dark conditions, all the other conditions remain the same. They noticed that antibacterial activity was not influenced by the presence or absence of light 32.
Growth feature of vibrios in microalgae culture. Lin et al. performed investigations to evaluate the effect of several microalgae species (i.e. Isochrysis galbana, Phaeodactylum tricornutum, Platymonas sp. and Chlorella sp.) strains in the growth of vibrios (V. parahaemolyticus, Vibrio spp, V. alginolyticus, and V. anguillarum) strains by using the batch culture method. They simultaneously conducted the control test of vibrios growth in sterilized seawater. Their results explained that vibrios grew well compared with the control test and that microalgae strain cultural systems successfully expel the vibrios. Additionally, they conclude that systems with long culture time present a decrease in the growth restriction ability. They finally suggest that the universal law that growth of vibrio is restricted by a cultural system of microalgae may exist 33.
Application of selected microalgae species for the inhibition of V. Parahaemolyticus bioactivity.
Based on the literature, we have considered testing the activity of seven different microalgae species (i.e. Chlamydomonas reinhardtii, Chlorella vulgaris, Klebsormidium flaccidum, Cosmarium impressulum, Kirchneriella lunaris, and Spirulina platensis) in the inhibition of V. parahaemolyticus. Mentioned microalgae have still not been tested for such vibrio strain or are barely studied. Procedure for evaluating the antibacterial activity is illustrated in Figure 2.
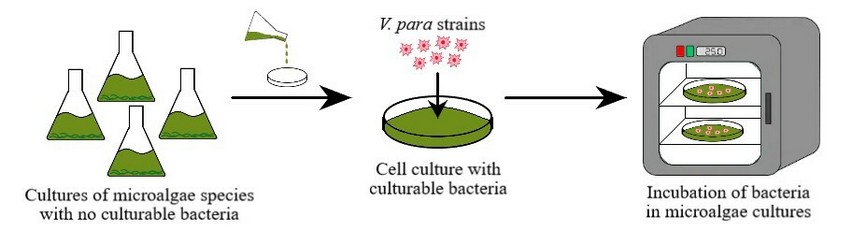
Figure 2. Proposed procedure to test the antibacterial activity of microalgae species against the V. parahaemolyticus.
Preparation of microalgae cultures. Cultures of microalgae species with no culturable bacteria need to be developed after inoculation of sterile seawater previously prepared, with microalgae-derived cells grown on a specific and suitable growth medium and temperature conditions (25°C) under continuous illumination. Every second day, cultures’ density has to be measured according to Kokou et al. 32
Phenotypical and growth tests. Once the cultures are pure, according to Smibert and Krieg, phenotypical tests must be conducted to determine Gram staining, fermentation, oxidase, and catalase test and susceptibility to vibriostatic agent 34. It is essential to depict the standard growth curve of each bacterial strain after culture in tryptic soy broth added NaCl solution (TSBS) for 24 hours. Colony forming units (CFU) can be enumerated by measuring optical density at 600 nm (OD600) as established by Makridis et al. 35
DNA extraction and purification. Recombinant DNA from the Vibrio parahaemolyticus can be readily purified, extracted and isolated by following standard procedure retrieved from the protocol presented in Roche Diagnosis 36. Polymerase Chain Reaction (PCR) procedures have to be performed to amplify our DNA samples. NanoDrop spectrophotometer assays should be conducted in order to quantify DNA samples.
CONCLUSIONS
Literature supports that V. parahaemolyticus is one of the causative agents of vibriosis in marine species such as shrimps, crabs, and fishes. Studies presented in this review have successfully measured the activity of this pathogen bacteria in the organisms suggesting that it may produce gastroenteritis and septicemia in humans by the ingestion of contaminated seafood, besides V. parahaemolyticus can markedly reduce the aquaculture industrial production resulting in great economic losses according to several types of researches. Most of the conventional solutions or methods are based on the use of commercial antibiotics or probiotics. However, these mentioned solutions have too many limitations, for instance, the excessive use of these compounds may lead to undesired and inadmissible side effects. Moreover, bacteria eventually develop antibiotic resistance making these treatments useless.
Multiple studies and investigations support the idea of the use of microalgae and their products, such as the biocapsules in the diet, in order to avoid the rate growth and proliferation of V. parahaemolyticus. All these procedures mentioned in the present review could be applied in the aquaculture to enhance and protect the average production of shrimps in Ecuador. Proposed microalgae-based solution using Chlamydomonas reinhardtii, Chlorella vulgaris, Klebsormidium flaccidum, Cosmarium impressulum, Kirchneriella lunaris, and Spirulina platensis may be a potential inhibitor of V. parahaemolyticus bioactivity. Hence, the use of microalgae species emerge as an environmentally friendly and cheaper solution against the V. parahaemolyticus bioactivity in marine organisms such as fish and shrimps and therefore can be applied in the aquaculture industrial field to improve yields and decrease costs.
REFERENCES
1. Egidius E. Vibriosis: Pathogenicity and pathology. A review. Aquaculture. 1987;67(1–2):15–28.
2. Egidius E, Wiik R, Andersen K, Hoff KA, Hjeltnes B. Vibrio salmonicida sp. nov., a New Fish Pathogen. Int J Syst Bacteriol. 2009;36(4):518–20.
3. Meeresuntersuchungen N, Meeresunters H. Vibrio diseases of marine fish populations. 1984;287:265–87.
4. Gomez-gil B. Vibriosis en camarones y su diagnóstico ( vibriosis in shrimp and its diagnosis ). 2016;(May).
5. Ina-Salwany MY, Al-saari N, Mohamad A, Mursidi FA, Mohd-Aris A, Amal MNA, et al. Vibriosis in Fish: A Review on Disease Development and Prevention. J Aquat Anim Health. 2019;31(1):3–22.
6. Karnjana K, Soowannayan C, Wongprasert K. Ethanolic extract of red seaweed Gracilaria fisheri and furanone eradicate Vibrio harveyi and Vibrio parahaemolyticus biofilms and ameliorate the bacterial infection in shrimp. Fish Shellfish Immunol. 2019;88:91–101.
7. Baker-Austin C, Stockley L, Rangdale R, Martinez-Urtaza J. Environmental occurrence and clinical impact of Vibrio vulnificus and Vibrio parahaemolyticus: A European perspective. Environ Microbiol Rep. 2010;2(1):7–18.
8. Cuéllar-Anjel J. Vibriosis Página 2 de 5. 2013;1–5.
9. LARSEN JL, PEDERSEN K, DALSGAARD I. Vibrio anguillarum serovars associated with vibriosis in fish. J Fish Dis. 1994;17(3):259–67.
10. Schiewe MH, Trust TJ, Crosa JH. Vibrio ordalii sp. nov.: A causative agent of vibriosis in fish. Curr Microbiol. 1981;6(6):343–8.
11. Vezzulli L, Brettar I, Pezzati E, Reid PC, Colwell RR, Höfle MG, et al. Long-term effects of ocean warming on the prokaryotic community: Evidence from the vibrios. ISME J. 2012;6(1):21–30.
12. Sung HH, Kou GH, Song YL. Vibriosis Resistance Induced by Glucan Treatment in Tiger Shrimp (Penaeus monodon). Fish Pathol. 2011;29(1):11–7.
13. Sotomayor MA, Balcázar JL. Inhibición de vibrios patógenos de camarón por mezclas de cepas probióticas Introducción Materiales y métodos. Investig Mar. 2003;9–15.
14. Zhao D, Application F, Data P, Examiner P, Mondesi RB, Examiner A, et al. Polyvalent attenuated live vacine for preventing and curing vibriosis of cultivated fish. 2010;2(12).
15. Dash P, Avunje S, Tandel RS, Sandeep KP, Panigrahi A. Biocontrol of Luminous Vibriosis in Shrimp Aquaculture: A Review of Current Approaches and Future Perspectives. Rev Fish Sci Aquac. 2017;25(3):245–55.
16. Van den HoeK C. Mann DG. Jahn HM. Algae An Introduction to phycology. Cambridge University Press. 1995.
17. Arvinda Swamy ML. Marine Algal Sources for Treating Bacterial Diseases. 1st ed. Vol. 64, Advances in Food and Nutrition Research. Elsevier Inc.; 2011. 71–84 p.
18. Anaya-Rosas RE, Rivas-Vega ME, Miranda-Baeza A, Piña-Valdez P, Nieves-Soto M. Effects of a co-culture of marine algae and shrimp (Litopenaeus vannamei) on the growth, survival and immune response of shrimp infected with Vibrio parahaemolyticus and white spot virus (WSSV). Fish Shellfish Immunol. 2019;87(2019):136–43.
19. Kasanah N, Amelia W, Mukminin A, Triyanto, Isnansetyo A. Antibacterial activity of Indonesian red algae Gracilaria edulis against bacterial fish pathogens and characterization of active fractions. Nat Prod Res. 2018;6419(May):1–5.
20. Silva GC, Albuquerque Costa R, Oliveira Peixoto JR, Pessoa Nascimento FE, Macedo Carneiro PB de, Fernandes Vieira RHS dos. Tropical Atlantic marine macroalgae with bioactivity against virulent and antibiotic resistant Vibrio. Lat Am J Aquat Res. 2013;41(1):183–8.
21. Widowati I, Zainuri M, Kusumaningrum HP, Maesaroh Y, Hardivillier Y, Leignel V, et al. Identification of agents causing vibriosis in litopenaeus vannamei shrimp culture in Kendal, Central Java, Indonesia and application of microalgae dunaliella salina and tetraselmis chui as bio-control agents against vibriosis. AACL Bioflux. 2018;11(1):101–7.
22. Lauritano C, Ferrante M, Rogato A. Marine Natural Products from Microalgae: An -Omics Overview. 2019;(17):1–18.
23. Wang R, Zhong Y, Gu X, Yuan J, Saeed AF, Wang S. The pathogenesis, detection, and prevention of Vibrio parahaemolyticus. Front Microbiol. 2015;6(MAR):1–13.
24. Park JC, Lee MS, Han DW, Lee DH, Park BJ, Lee IS, et al. Inactivation of Vibrio parahaemolyticus in Effluent Seawater by Alternating-Current Treatment. Appl Environ Microbiol. 2004;70(3):1833–5.
25. Wang JJ, Sun WS, Jin MT, Liu HQ, Zhang W, Sun XH, et al. Fate of Vibrio parahaemolyticus on shrimp after acidic electrolyzed water treatment. Int J Food Microbiol. 2014;179:50–6.
26. Mahmoud BSM. Effect of X-ray treatments on inoculated Escherichia coli O157: H7, Salmonella enterica, Shigella flexneri and Vibrio parahaemolyticus in ready-to-eat shrimp. Food Microbiol. 2009;26(8):860–4.
27. Immanuel G, Vincybai VC, Sivaram V, Palavesam A, Marian MP. Effect of butanolic extracts from terrestrial herbs and seaweeds on the survival, growth and pathogen (Vibrio parahaemolyticus) load on shrimp Penaeus indicus juveniles. Aquaculture. 2004;236(1–4):53–65.
28. Li SS, Tsai HJ. Transgenic microalgae as a non-antibiotic bactericide producer to defend against bacterial pathogen infection in the fish digestive tract. Fish Shellfish Immunol. 2009;26(2):316–25.
29. KUMAZAWA NH, FUKUMA N, KOMODA Y. Attachment of Vibrio parahaemolyticus strains to estuarine algae. J Vet Med Sci. 2011;53(2):201–5.
30. Molina-Cárdenas CA, Sánchez-Saavedra M del P, Lizárraga-Partida ML. Inhibition of pathogenic Vibrio by the microalgae Isochrysis galbana. J Appl Phycol. 2014;26(6):2347–55.
31. Sudha SS, Rajamanickam K, Rengaramanujam J. Microalgae mediated synthesis of silver nanoparticles and their antibacterial activity against pathogenic bacteria. Indian J Exp Biol. 2013;51(5):393–9.
32. Kokou F, Makridis P, Kentouri M, Divanach P. Antibacterial activity in microalgae cultures. Aquac Res. 2012;43(10):1520–7.
33. Lin W, Chen D, Lin X. Growth feature of vibrios in microalgae cultural systems. Oceanol Limnol Sin. 2000;(31):4.
34. Smibert R.M. & Krieg N.R. Phenotypic characterization. Methods Gen Mol Bacteriol. 1994;607–53.
35. Makridis P, Jon Fjellheim A, Skjermo J, Vadstein O. Colonization of the gut in first feeding turbot by bacterial strains added to the water or bioencapsulated in rotifers. Aquac Int. 2000;8(5):367–80.
36. Roche Diagnostics Group. High Pure PCR Template Preparation Kit Rapidly purify genomic DNA for diverse applications. 2008.
Received: 17 April, 2019
Accepted: 23 May 2019
Freddy Figueroa, Bryan Aldaz
School of Biological Sciences and Engineering, Yachay Tech University
